Challenges of Hepatitis B Virus Reactivation and CD19 Testing Following Tafasitamab Plus Lenalidomide for Relapsed Diffuse Large B-Cell Lymphoma
DOI:
https://doi.org/10.14740/jmc5313Keywords:
Diffuse large B-cell lymphoma, Hepatitis B virus, Tafasitamab, LenalidomideAbstract
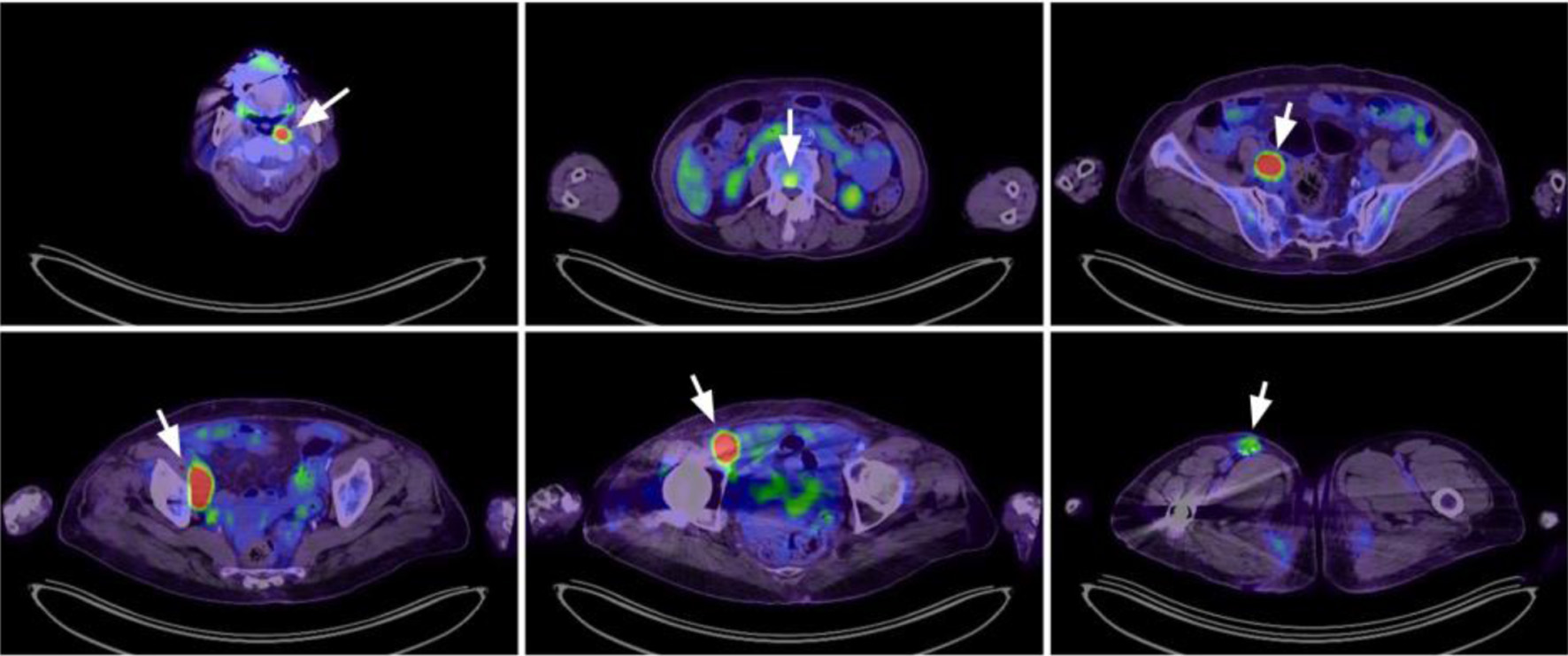
The outcomes of transplant-ineligible patients with relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL) are generally poor but much improved by novel targeted therapies and immunotherapies. Tafasitamab plus lenalidomide is an approved regimen for patients with R/R DLBCL. Nonetheless, the clinical data regarding Asian populations and hepatitis B virus (HBV) carriers are lacking in both the pivotal L-MIND and real-world studies. We present an 81-year-old HBV carrier with relapsed DLBCL, who achieved and continued a complete metabolic response (CMR) during 1.5 years of tafasitamab plus lenalidomide as second-line therapy. Notably, an episode of HBV reactivation occurred after four cycles of tafasitamab plus lenalidomide, which was early detected and successfully managed with preemptive nucleotide analogues. Interestingly, CD19 was not detectable by flow cytometry after 10 cycles of treatment despite continuous control of the disease. CD19 expression may be diminished by exams using a tafasitamab-competitive-binding clone. This case highlights not only the concern of HBV reactivation, but also the diagnostic challenge due to CD19 epitope masking following tafasitamab therapy.
Published
Issue
Section
License
Copyright (c) 2026 The authors

This work is licensed under a Creative Commons Attribution 4.0 International License.









