| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://jmc.elmerpub.com |
Case Report
Volume 17, Number 6, June 2026, pages 271-274
A Case Series of Late Myocardial Infarction Following Self-Expanding Transcatheter Aortic Valve Replacement
Muhammad Saada, d, Mohammad Hashim Jilanib, Mohsin Munawarb, Cristiano Spadaccioc, Rishi Sukhijab, Arif Imranb
aDepartment of Internal Medicine, The Jewish Hospital of Cincinnati, Cincinnati, OH, USA
bDivision of Cardiology, University of Cincinnati School of Medicine, Cincinnati, OH, USA
cDepartment of Cardiothoracic Surgery, University of Cincinnati School of Medicine, Cincinnati, OH, USA
dCorresponding Author: Muhammad Saad, Department of Internal Medicine, The Jewish Hospital of Cincinnati, OH 45236, USA
Manuscript submitted February 10, 2026, accepted April 6, 2026, published online April 29, 2026
Short title: Late Embolic MI After Self-Expanding TAVR
doi: https://doi.org/10.14740/jmc5303
| Abstract | ▴Top |
Embolic stroke is a recognized complication of transcatheter aortic valve replacement (TAVR); however, coronary embolism is rarely reported, particularly when occurring late after valve implantation. We described three patients presenting with ST-segment elevation myocardial infarction (STEMI) between 10 and 50 months after self-expanding TAVR. All patients had angiographically normal coronary arteries prior to TAVR, severe native aortic valve calcification, and well-controlled cardiovascular risk factors, and were maintained on guideline-directed single antiplatelet therapy. At presentation, coronary angiography demonstrated abrupt 100% coronary occlusion without angiographic evidence of underlying atherosclerotic disease. Aspiration thrombectomy was required in all three cases. These cases illustrate a rare presentation of late myocardial infarction following self-expanding TAVR with angiographic features possibly suggestive of a non-atherosclerotic mechanism. Although a definitive embolic source cannot be established, delayed embolization of calcific or thrombotic material may represent a plausible explanation. These observations are hypothesis-generating and underscore the need for clinical vigilance and further mechanistic and imaging-based studies to better characterize potential late thromboembolic pathways after TAVR.
Keywords: TAVR; Acute myocardial infarction; Aortic stenosis; Transcatheter; Aspiration thrombectomy
| Introduction | ▴Top |
Aortic stenosis (AS) is one of the most prevalent valvular heart diseases [1] with a high mortality rate of up to 50% at 2 years in symptomatic patients [2]. Aortic valve replacement, either transcatheter aortic valve replacement (TAVR) or surgical aortic valve replacement (SAVR), is the treatment of choice for severe symptomatic AS.
TAVR is becoming increasingly common, supported by trials such as PARTNER 3 and EVOLUT, which have demonstrated that TAVR is either superior or at least non-inferior to SAVR [3]. Initially, TAVR was reserved for high-risk patients who were deemed unsafe for surgical replacement, but now it is approved for aortic valve replacement in patients with symptomatic AS, including those at high, intermediate, or low risk [4]. In TAVR, two types of valves are widely used: self-expanding valves (SEVs) and balloon-expandable valves (BEVs) [5]. Paravalvular leak (PVL), conduction disturbances, and stroke are some well-known complications of TAVR.
Systemic embolism, causing stroke (1–2% incidence), is a well-documented early periprocedural complication. According to current American and European guidelines, lifelong single antiplatelet therapy (SAPT) with aspirin is recommended after TAVR unless there is another indication for dual antiplatelet therapy (DAPT) or anticoagulation [6, 7].
We present a case series of three patients who developed late ST-segment elevation myocardial infarction (STEMI) due to abrupt coronary occlusion several months to years after self-expanding TAVR, highlighting a rare clinical phenotype and raising hypotheses regarding potential mechanisms.
| Case Reports | ▴Top |
Case 1
A 66-year-old man with severe bicuspid AS underwent TAVR with a 34-mm self-expanding Evolut PRO+ valve. The native aortic valve was severely calcified with an Agatston calcium score of 2,443. Balloon valvuloplasty and post-dilation were performed during the TAVR procedure. The patient was discharged on lifelong SAPT with aspirin.
Fifty months after TAVR, he presented with anterolateral STEMI. Notably, the pre-TAVR coronary angiogram demonstrated normal coronary arteries without evidence of obstructive coronary artery disease (CAD) (Fig. 1a); however, repeat left heart catheterization at the time of presentation with STEMI revealed abrupt 100% occlusion of the left anterior descending artery (Fig. 1b), diagonal branch (Fig. 1b), and obtuse marginal (OM) branch (Fig. 1c), consistent with an embolic pattern. Aspiration thrombectomy restored TIMI 3 flow in all the affected vessels (Fig. 1d, e).
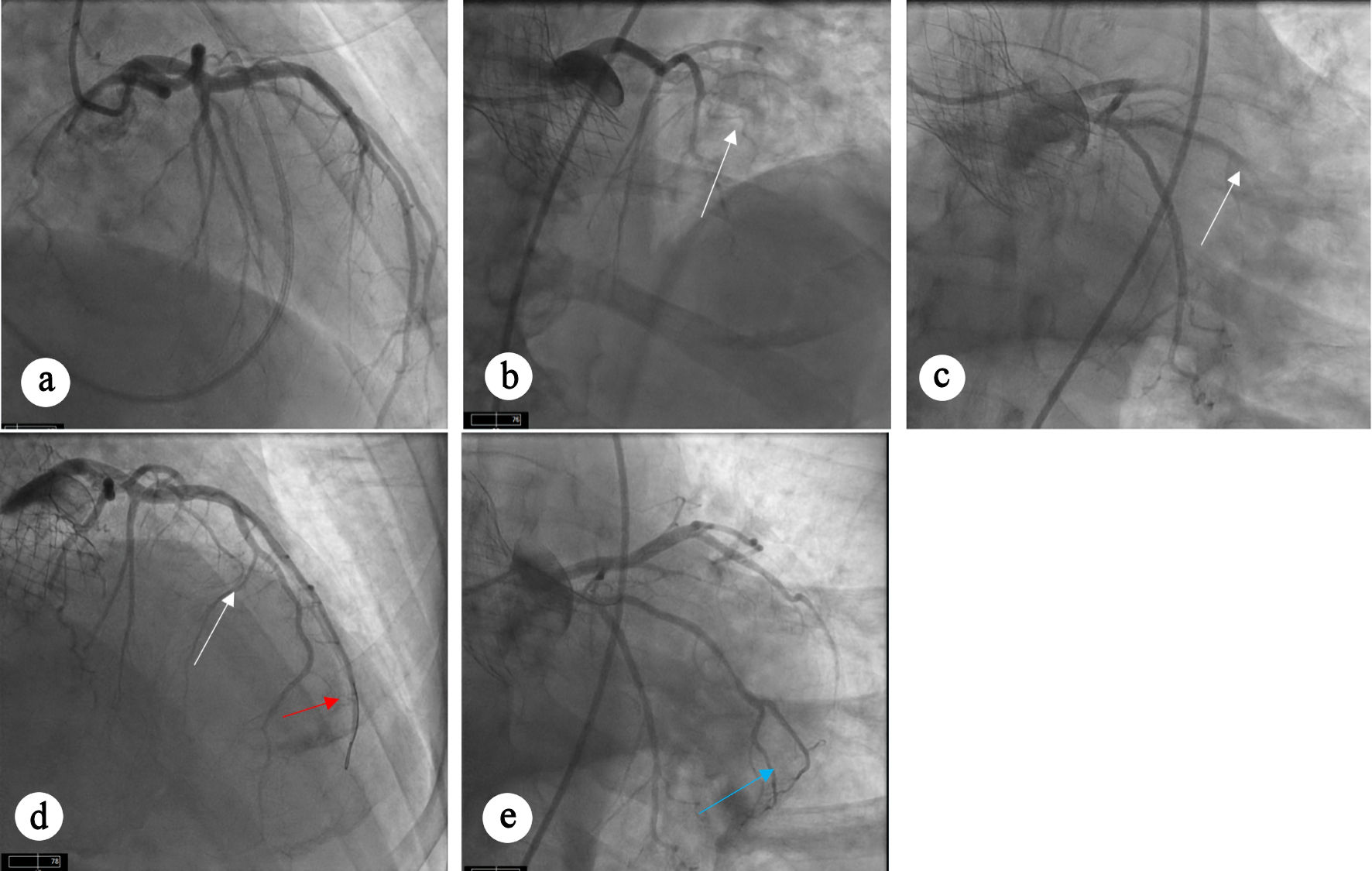 Click for large image | Figure 1. Angiographic views showing coronary arteries. (a) Diagnostic angiogram before TAVR procedure showing angiographically normal coronary arteries without any evidence of obstructive CAD. (b) Occlusion of LAD and diagonal branch artery. (c) Occlusion of OM artery. (d, e) Restoration of flow can be seen in LAD (white arrow), diagonal branch (red arrow), and OM artery (blue arrow) after aspiration thrombectomy. CAD: coronary artery disease; LAD: left anterior descending; OM: obtuse marginal; TAVR: transcatheter aortic valve replacement. |
Case 2
A 79-year-old man with severe trileaflet AS underwent TAVR using a 29-mm Evolut FX valve. Native valve calcium score was 2,972. Balloon valvuloplasty was performed without post-dilation. He received lifelong aspirin monotherapy.
Ten months after TAVR, the patient presented with inferolateral STEMI. Coronary angiography demonstrated abrupt 100% occlusion of the two OM branches (Fig. 2a). Aspiration thrombectomy resulted in restoration of TIMI 3 flow in both OM branches (Fig. 2b). Review of pre-TAVR coronary angiogram confirmed previously patent vessels without any evidence of obstructive CAD (Fig. 2c).
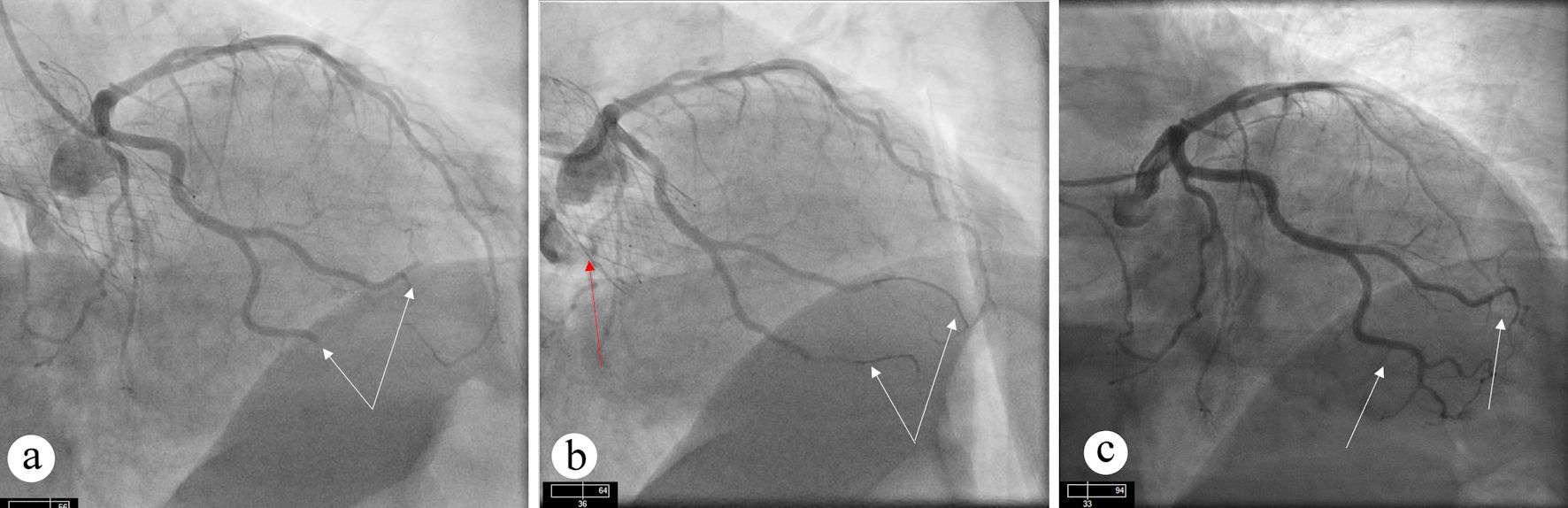 Click for large image | Figure 2. Angiographic views showing occlusion and restoration of flow after intervention. (a) Occlusion of OM branches (white arrows) can be seen. (b) Restoration of flow can be seen in OM branches (white arrow) after aspiration thrombectomy. TAVR valve (red arrow) can also be appreciated. (c) Diagnostic angiogram before TAVR procedure showing patent OM branches. OM: obtuse marginal; TAVR: transcatheter aortic valve replacement. |
Case 3
An 80-year-old man with severe trileaflet AS underwent TAVR with a 29-mm Evolut PRO+ valve. The native valve calcium score was 3,422, and no balloon valvuloplasty or post-dilation was performed during the TAVR procedure. The patient was discharged on aspirin monotherapy.
Twenty-nine months later, he presented with inferolateral STEMI. Coronary angiography showed complete occlusion of an OM branch (Fig. 3a). Despite multiple attempts with balloon angioplasty and aspiration thrombectomy, adequate coronary flow could not be restored (Fig. 3b). Notably, the pre-TAVR coronary angiogram demonstrated patent OM branches without any evidence of obstructive CAD (Fig. 3c).
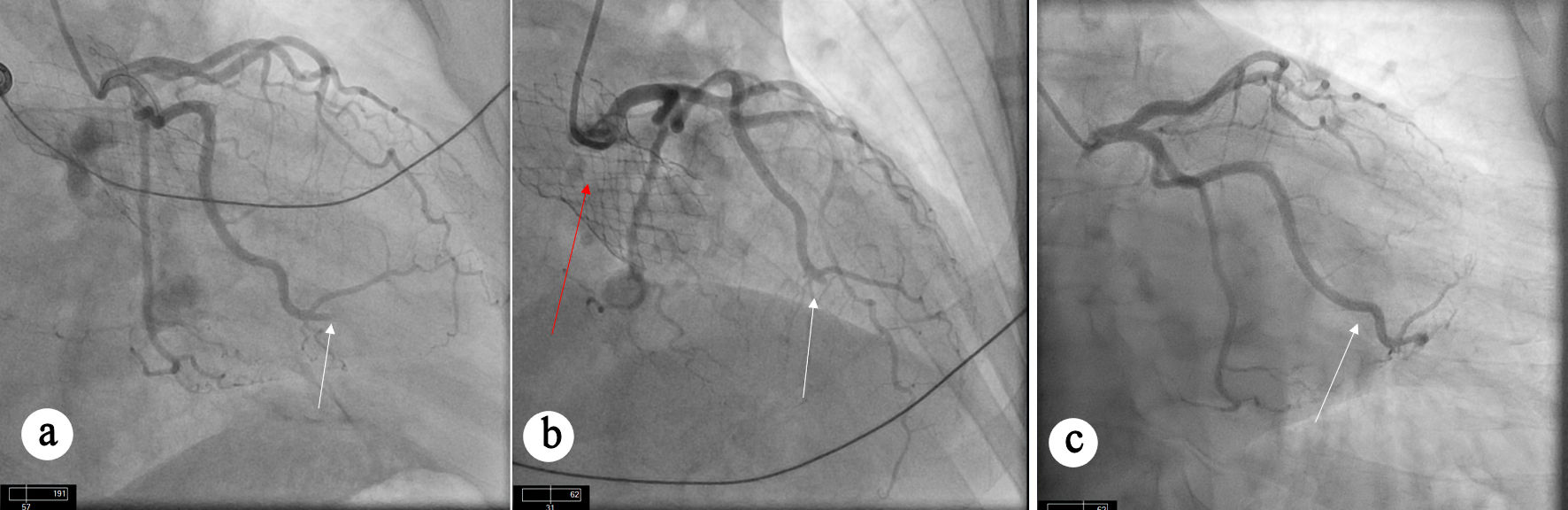 Click for large image | Figure 3. Angiographic views showing persistent occlusion of OM branch. (a) Occlusion of OM artery (white arrow) can be seen. (b) Persistent occlusion of OM artery (white arrow) despite aspiration thrombectomy. TAVR valve (red arrow) can also be seen. (c) Diagnostic angiogram before TAVR procedure showing patent OM artery without obstructive CAD. CAD: coronary artery disease; OM: obtuse marginal; TAVR: transcatheter aortic valve replacement. |
| Discussion | ▴Top |
STEMI following TAVR is associated with worse clinical outcomes and higher short- and long-term mortality, likely attributable to the advanced comorbidity burden of this patient population [8, 9]. Although non-atherothrombotic mechanisms such as coronary embolism have been described in the literature [8, 10, 11], their precise contribution and clinical significance in TAVR patients remain incompletely understood. Our case series highlights a distinct angiographic pattern of late STEMI occurring in patients without prior obstructive CAD, characterized by abrupt coronary occlusion requiring aspiration thrombectomy in the setting of minimal atherosclerotic plaque. These features raise the possibility of an embolic mechanism. Supporting this hypothesis, a recent case report documented histopathologically confirmed calcific coronary embolization as a cause of STEMI following TAVR; however, this finding was associated with the ACURATE neo2 M valve [12].
Several mechanisms may plausibly account for these findings, including delayed embolization of calcific debris from severely calcified native valves, subclinical leaflet thrombosis, or valve-related flow disturbances. Importantly, these events occurred despite adherence to guideline-directed SAPT and should not be interpreted as justification for escalation of routine antithrombotic therapy, which is contraindicated in current European guidelines [7]. Alternative explanations cannot be completely excluded. Plaque erosion, occult atrial arrhythmias, or in-situ coronary thrombosis remain possible given the absence of intravascular imaging, histopathologic analysis, or contemporary cardiac computed tomography. Accordingly, these observations should be interpreted as hypothesis-generating rather than evidence of a causal relationship between TAVR and late coronary embolic events.
This case series has several limitations, including a small sample size, retrospective design, and lack of advanced imaging or histopathologic confirmation of embolic material. Incomplete procedural anatomic data, such as coronary heights and valve implantation depth, further limit mechanistic inference.
Taken together, STEMI occurring late after self-expanding TAVR appears to be uncommon but associated with worse outcomes [8]. It may plausibly reflect a non-atherosclerotic process in selected patients with low traditional cardiovascular risks and angiographic findings not suggestive of plaque rupture. Intravascular imaging, rhythm monitoring, and histopathological and mechanistic studies are needed to better characterize the underlying etiology of STEMI in this population and potentially inform management strategies.
Conclusions
Late myocardial infarction after TAVR is rare but, in certain patients, may plausibly represent a non-atherosclerotic process occurring despite adherence to guideline-directed antiplatelet therapy. In patients with low baseline atherosclerotic risk factors, absence of atrial arrhythmias or intracardiac thrombus, and coronary angiography demonstrating abrupt vessel occlusion without underlying atherosclerotic disease, an embolic mechanism may be considered as a potential explanation. Intravascular imaging and histopathological evaluation, when feasible, may provide valuable insights into lesion characterization. Severe native aortic valve calcification may represent a theoretical substrate for delayed embolic phenomena after TAVR; however, a direct causal relationship cannot be established based on the current observations. These findings are exploratory and hypothesis-generating and should not be interpreted as evidence to modify current guideline-recommended antithrombotic strategies.
Acknowledgments
The authors acknowledge all healthcare professionals and staff who contributed to the care of the patients described in this case series. The authors appreciate the collaborative efforts that supported the completion of this work.
Financial Disclosure
None to declare.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Informed Consent
Informed consent was obtained.
Author Contributions
Muhammad Saad, Mohammad Hashim Jilani, Mohsin Munawar performed the literature review, collected the data, and wrote the manuscript. Cristiano Spadaccio, Rishi Sukhija, Arif Imran performed the procedures, reviewed the manuscript, and contributed to the discussion.
Data Availability
The authors declare that data supporting the findings of this study are available within the article. Additional information is available from the corresponding author upon reasonable request.
Abbreviations
AS: aortic stenosis; BEV: balloon expandable valve; CAD: coronary artery disease; DAPT: dual antiplatelet therapy; LAD: left anterior descending; MI: myocardial infarction; OM: obtuse marginal; PVL: paravalvular leak; SAPT: single antiplatelet therapy; SAVR: surgical aortic valve replacement; SEV: self-expanding valve; STEMI: ST-segment elevation myocardial infarction; TAVR: transcatheter aortic valve replacement
| References | ▴Top |
- Kanwar A, Thaden JJ, Nkomo VT. Management of patients with aortic valve stenosis. Mayo Clin Proc. 2018;93(4):488-508.
doi pubmed - Iervolino A, Singh SSA, Nappi P, Bellomo F, Nappi F. Percutaneous versus surgical intervention for severe aortic valve stenosis: a systematic review. Biomed Res Int. 2021;2021:3973924.
doi pubmed - Itchhaporia D. TAVR 20 years later: a story of disruptive transformation. J Am Coll Cardiol. 2022;79(13):1314-1316.
doi pubmed - Abusnina W, Machanahalli Balakrishna A, Ismayl M, Latif A, Reda Mostafa M, Al-Abdouh A, Junaid Ahsan M, et al. Comparison of Transfemoral versus Transsubclavian/Transaxillary access for transcatheter aortic valve replacement: A systematic review and meta-analysis. Int J Cardiol Heart Vasc. 2022;43:101156.
doi pubmed - Vlastra W, Chandrasekhar J, Munoz-Garcia AJ, Tchetche D, de Brito FS, Jr., Barbanti M, Kornowski R, et al. Comparison of balloon-expandable vs. self-expandable valves in patients undergoing transfemoral transcatheter aortic valve implantation: from the CENTER-collaboration. Eur Heart J. 2019;40(5):456-465.
doi pubmed - Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP, 3rd, Gentile F, Jneid H, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143(5):e35-e71.
doi pubmed - Praz F, Borger MA, Lanz J, Marin-Cuartas M, Abreu A, Adamo M, Ajmone Marsan N, et al. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2025;46(44):4635-4736.
doi pubmed - Faroux L, Lhermusier T, Vincent F, Nombela-Franco L, Tchetche D, Barbanti M, Abdel-Wahab M, et al. ST-segment elevation myocardial infarction following transcatheter aortic valve replacement. J Am Coll Cardiol. 2021;77(17):2187-2199.
doi pubmed - Gupta T, Zimmer J, Lahoud RN, Murphy HR, Harris AH, Kolte D, Hirashima F, et al. National trends and outcomes of acute myocardial infarction after transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2024;17(10):1267-1276.
doi pubmed - Akuka A, Landes U, Manevich L, Rubinshtein R, Danenberg HD. Coronary embolism after transcatheter aortic valve replacement-case series and review of literature. Am J Cardiol. 2023;205:234-240.
doi pubmed - Raphael CE, Heit JA, Reeder GS, Bois MC, Maleszewski JJ, Tilbury RT, Holmes DR, Jr. Coronary embolus: an underappreciated cause of acute coronary syndromes. JACC Cardiovasc Interv. 2018;11(2):172-180.
doi pubmed - Ene C, Vandeloo B, Billen M, Lefesvre P, Argacha JF. Late coronary embolisation after transcatheter aortic valve implantation. EuroIntervention. 2025;21(2):e140-e141.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, including commercial use, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.