| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://jmc.elmerpub.com |
Case Report
Volume 16, Number 8, August 2025, pages 300-308
Extended-Hours Hemodialysis Without Dietary Restrictions Reduces Risk of Vascular Calcification
Hiroshi Kanedaa, Toshiro Nishiyamab, d, Kazunori Owadaa, Koichi Katayosea, Yutaka Takagia, Katsuhiko Suzukic, Fumika Kanedaa
aDepartment of Nephrology, Kamome Clinic, Iwaki City, Fukushima 972-8301,
Japan
bDepartment of Medical Engineering, Kamome Clinic, Iwaki City, Fukushima
972-8301, Japan
cDepartment of Nephrology, Nagoya University Graduate School of
Medicine, Nagoya City 466-8550, Japan
dCorresponding Author: Toshiro Nishiyama,
Department of Medical Engineering, Kamome Clinic, Iwaki City, Fukushima 972-8301, Japan
Manuscript submitted May 26, 2025, accepted August 6, 2025, published online August 22,
2025
Short title: Prevent Vascular Calcification in Dialysis Patients
doi:
https://doi.org/10.14740/jmc5146
| Abstract | ▴Top |
Vascular calcification is common in dialysis patients and is severely associated with cardiovascular morbidity and mortality. They have mineral metabolism disorders, which are considered to promote vascular calcification. In addition, hypertension and malnutrition, both prevalent in dialysis patients, are also considered risk factors contributing to vascular calcification. Our clinic has implemented extended-hours hemodialysis without dietary restrictions to solve the problems of hypertension and malnutrition in dialysis patients. We report a case where aortic calcification was slight despite 34 years of long-term dialysis. He underwent his first computed tomography (CT) scan of the chest and abdomen 29 years after initiating hemodialysis to evaluate his kidney transplant. The abdominal aortic calcification index 29 years after initiating dialysis was low at 5.8%, and even after 5 years, it was only 6.7%. The coronary artery calcification score was moderate at 214.0, but the degree of vascular calcification appeared milder than in conventional dialysis patients. His dialysis modality is unique. He began extended-hours hemodialysis without dietary restrictions 1.5 years after his transfer to our clinic, extending his dialysis time to 6 h. He gradually increased it to 10 h, and the dietary restriction was significantly relaxed. Moreover, he has been undergoing this treatment for about 26 years. His muscle mass increased, and his nutritional status remained adequate. The mean serum phosphorus and calcium levels were within the normal range (5.4 ± 0.5 mg/dL and 8.8 ± 0.3 mg/dL), and the calcium-based phosphate binder was successfully reduced and eventually discontinued. Furthermore, the blood pressure normalized without the use of antihypertensive medications. These may have removed some of the risk factors for vascular calcification. Recent studies suggest that extended-hours hemodialysis without dietary restrictions significantly lowers calciprotein particle levels, a risk factor for vascular calcification, compared to conventional dialysis. In conclusion, extended-hours hemodialysis without dietary restrictions may reduce the risk of vascular calcification by improving control of malnutrition, hypertension, and mineral metabolism.
Keywords: Vascular calcification; Extended-hours hemodialysis without dietary restrictions; Mineral metabolism; Malnutrition; Calciprotein particles
| Introduction | ▴Top |
Vascular calcification is prevalent in chronic kidney disease, particularly in dialysis patients, and is strongly associated with cardiovascular morbidity and mortality due to vascular calcification [1, 2]. Vascular calcification also occurs in the general population, but the classic risk factors are old age, male, diabetes mellitus, hypertension, and dyslipidemia. In contrast, known risk factors for vascular calcification in dialysis patients include disorders of mineral metabolism, such as elevated serum phosphate and calcium levels, uremia, inflammation, and nutritional disorders [3].
In recent years, there has been a focus on the association between calciprotein particles (CPPs), circulating particles in the blood formed by the binding of calcium phosphate and protein (fetuin-A), and vascular calcification [4, 5]. It has been reported that CPP levels are elevated in dialysis patients, leading to early progression of vascular calcification [6]. In addition, several reports have shown that malnutrition and inflammation are closely associated with vascular calcification in dialysis patients [7, 8]. Dialysis patients are usually obliged to restrict their diet to control serum phosphate levels. Many are malnourished, and reduced protein and calorie intake due to dietary restrictions leads to more significant mortality [9].
The dialysis treatment modality in our clinic is unique, with two main strategies. First, the clinic specializes in extended-hours hemodialysis. Patients undergo at least 6 h of hemodialysis treatment per session. In addition, we recommend that patients undergo extended hemodialysis for up to 8 h. Second, we recommend that they eat the same diet as their healthy family members instead of imposing dietary restrictions. We have been implementing this treatment for 26 years, and extended-hours hemodialysis has been reported to improve nutritional status and hypertension [10-13]. Herein, we report a case of a patient with slight aortic calcification despite 34 years of long-term hemodialysis.
| Case Report | ▴Top |
A 58-year-old man initiated hemodialysis at the age of 24 years at a general hospital due to end-stage renal failure caused by congenital hydronephrosis. The conventional dialysis program was three times a week for 4 h, but from the beginning of induction, it was extended to three times a week for 5 h. The patient was transferred to our clinic at the age of 31 years, 6.5 years after initiating dialysis. Initially, our facility provided 5-h post-dilution offline hemodiafiltration with a substitution volume of 6 L per session, administered three times per week. After 1.5 years, extended-hours hemodiafiltration was started. The patient opted for 6-h sessions due to work-related circumstances. With the dialysis duration now 1.5 times longer than that of conventional treatment, dietary restrictions have become less stringent. Extended-hours hemodiafiltration was continued for 7.5 years before transitioning to nocturnal hemodialysis. Our clinic began a 2.5-year trial of in-center nocturnal hemodialysis 9 years after his transfer (39 years old). There, he underwent nocturnal hemodialysis for 8 h per session. After the center’s nocturnal hemodialysis program finished, he returned to 6-h dialysis; however, he extended to 7-h dialysis 14.8 years later (45 years old). Subsequently, the dialysis time was extended further, and the dietary restriction was significantly relaxed and no longer imposed. After 23.6 years (54 years old), he was on extended-hours hemodialysis for 10 h at his request. However, he has continued his duties without issues by utilizing flexible working hours through cooperation with his workplace. He had been undergoing extended-hours hemodialysis for approximately 26 years (Fig. 1). His hemodialysis treatment was unique and different from conventional hemodialysis.
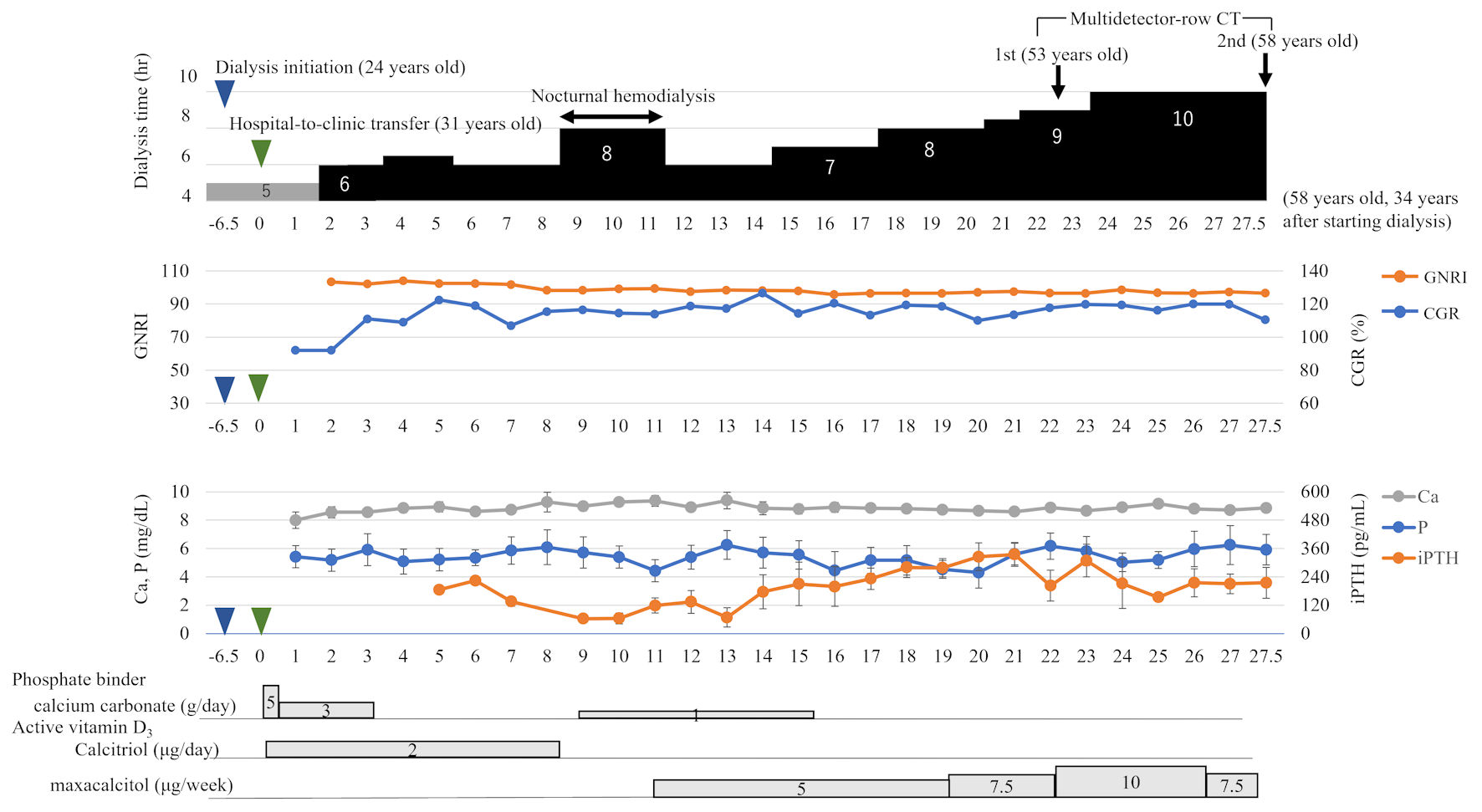 Click for large image |
Figure 1. Clinical course over 27.5 years (31 - 58 years old) after the transfer. The patient began 6-h extended-hours hemodialysis without dietary restrictions 1.5 years after the transfer. The dialysis time was gradually extended to 10 h, and extended-hours hemodialysis without dietary restrictions has been maintained for 26 years. The GNRI levels, a nutritional index, remained in the normal range. The CGR, a muscle mass index, increased after the change in dialysis time to 6 h, reaching 109% after 3 years. Calcium remained slightly low, phosphorus was almost in the normal range, and the phosphate binder calcium carbonate was discontinued after 16 years. After 17 years, the iPTH levels showed an upward trend, while the active vitamin D3 dosage increased. The blue arrow indicates the dialysis initiation, and the green arrow indicates general hospital-to-clinic transfer. GNRI: geriatric nutritional risk index; CGR: creatinine generation rate; Ca: calcium; P: phosphate; iPTH: intact parathyroid hormone; CT: computed tomography. |
He underwent his first chest and abdominal computed tomography (CT) scan as a screening test for kidney transplant donor registration 29 years after initiated hemodialysis (53 years old). A second CT scan was performed 5 years later (58 years old). The Aquilio 64 (Canon Medical Systems Corporation, Tokyo, Japan) was used for CT scanning. Interestingly, vascular calcification was only slight, even 29 years after initiating hemodialysis. Furthermore, CT scan performed 5 years later (34 years later) showed almost no change in vascular calcification (Figs. 2, 3). In addition, vascular calcification was quantified morphometrically using CT slices. The abdominal aorta was used to assess vascular calcification, and the abdominal aortic calcification index (AACI) was examined as a clinical index [14]. AACI (%) was measured by quantitatively assessing 10 slices of the abdominal aorta scanned at 10-mm intervals from the common iliac artery bifurcation. The cross-section of the abdominal aorta in each slice was radially divided into 12 sectors, and the number of sectors with calcifications in each slice was counted. The number of sectors with calcification in each slice was divided by 12, and the values for 10 slices were summed. The total was divided by 10, and the number of slices examined was averaged and multiplied by 100 to express the percentage [15]. The AACI was calculated using the following formula:
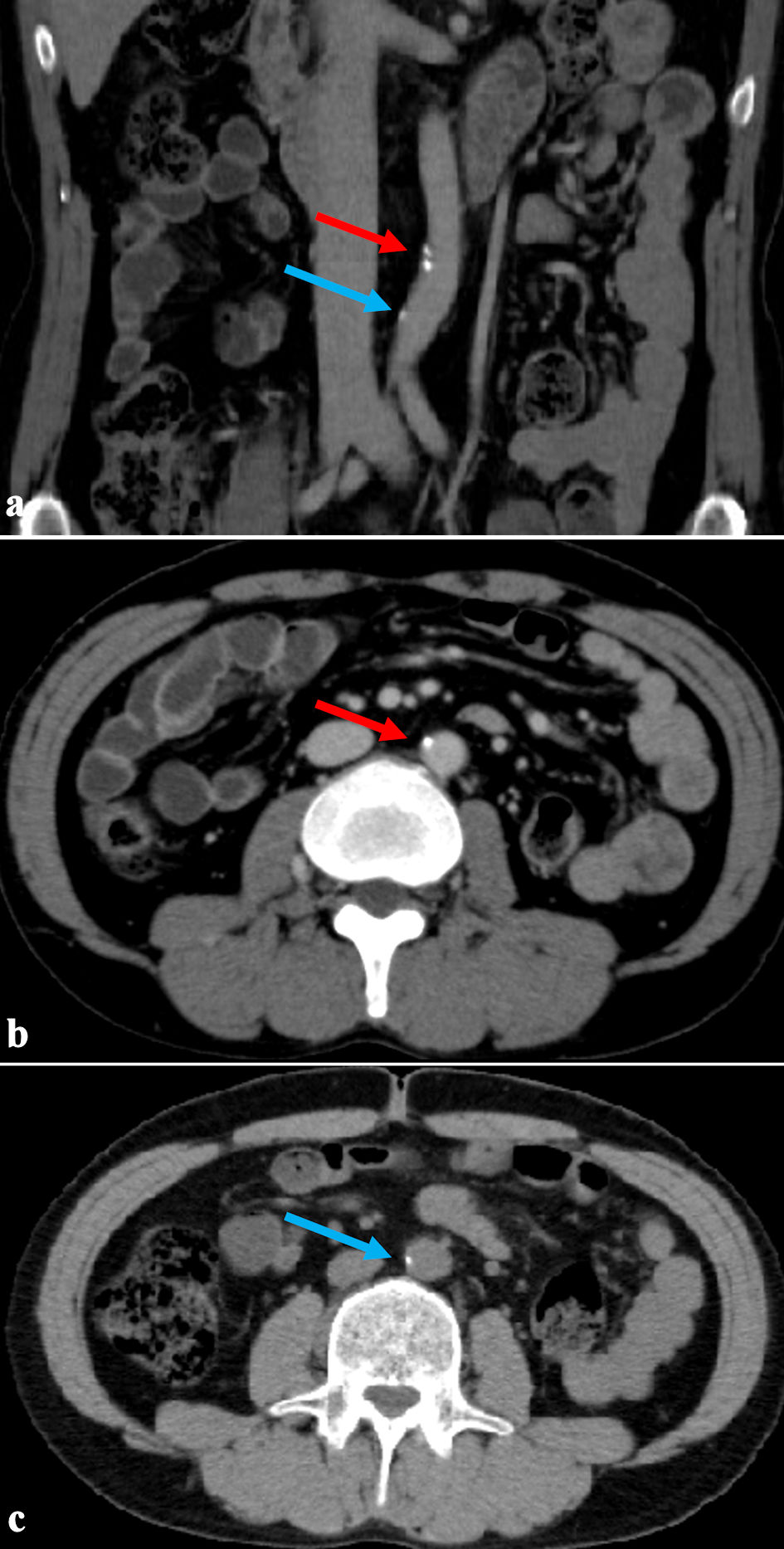 Click for large image |
Figure 2. The first abdominal CT scan 29 years after the initiation of dialysis (age 53). (a) Coronal section. (b, c) Axial sections showing abdominal aortic calcification area. The red arrow indicates the upper calcification area, and the blue arrow indicates the lower area. Despite undergoing 29 years of long-term dialysis, calcification remained slight. CT: computed tomography. |
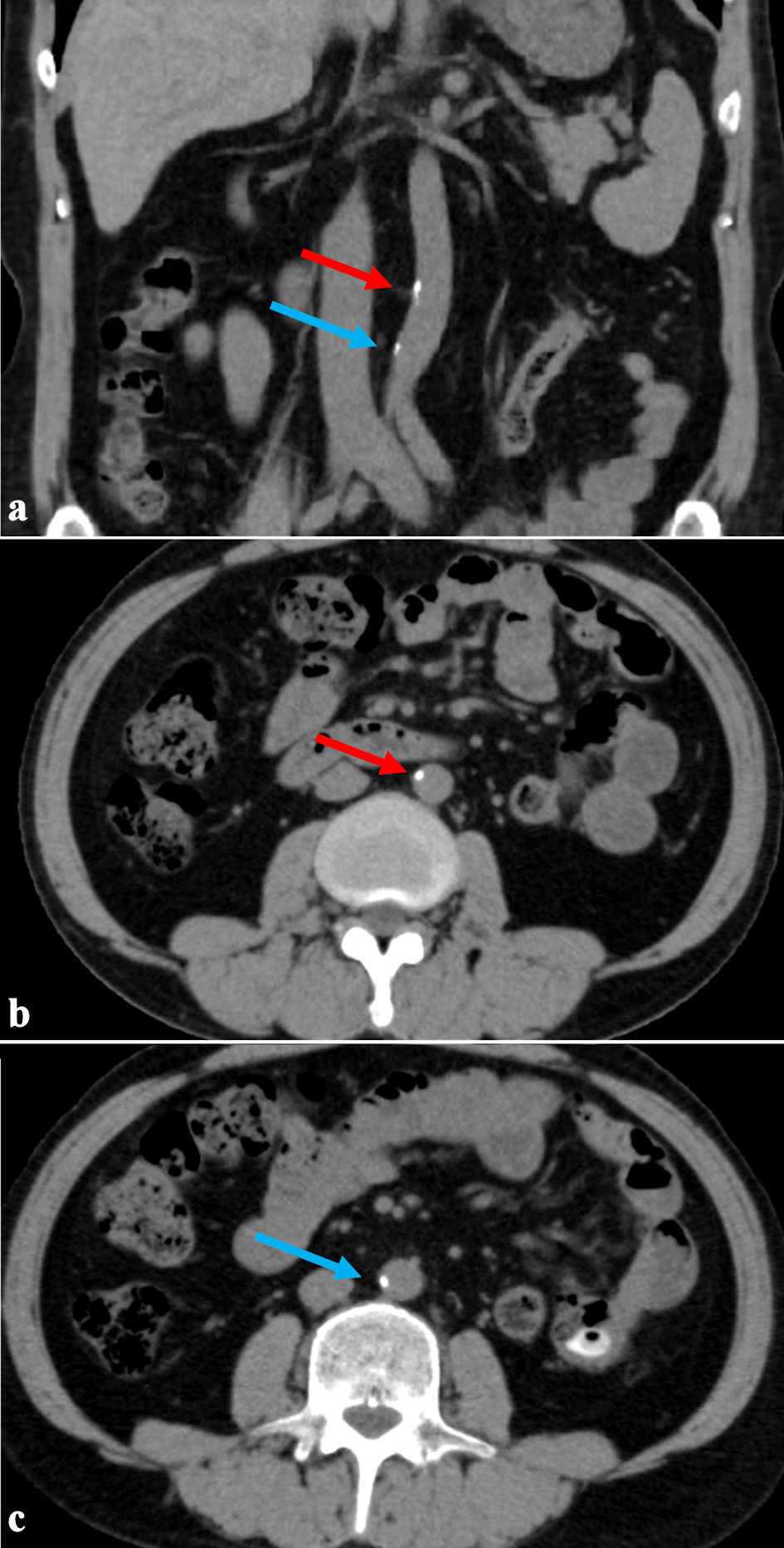 Click for large image |
Figure 3. The second abdominal CT scan taken 34 years after initiating dialysis (5 years after the first CT scan). (a) Coronal section. (b, c) Axial sections showing abdominal aortic calcification area. The red arrow indicates the upper calcification area, and the blue arrow indicates the lower area. There has been little change in the calcified lesions after 5 years. CT: computed tomography. |
A CT scan performed 29 years after initiating hemodialysis revealed vascular calcification in the abdominal aorta; however, the AACI was low at 5.8%. Five years later, the AACI increased slightly to 6.7%; however, no new vascular calcifications were observed in the thoracoabdominal aorta during these 5 years (Fig. 3b, c). However, calcification of the internal iliac artery was found, but there was no evidence of buttock claudication due to impaired blood flow in the internal iliac artery (Fig. 4).
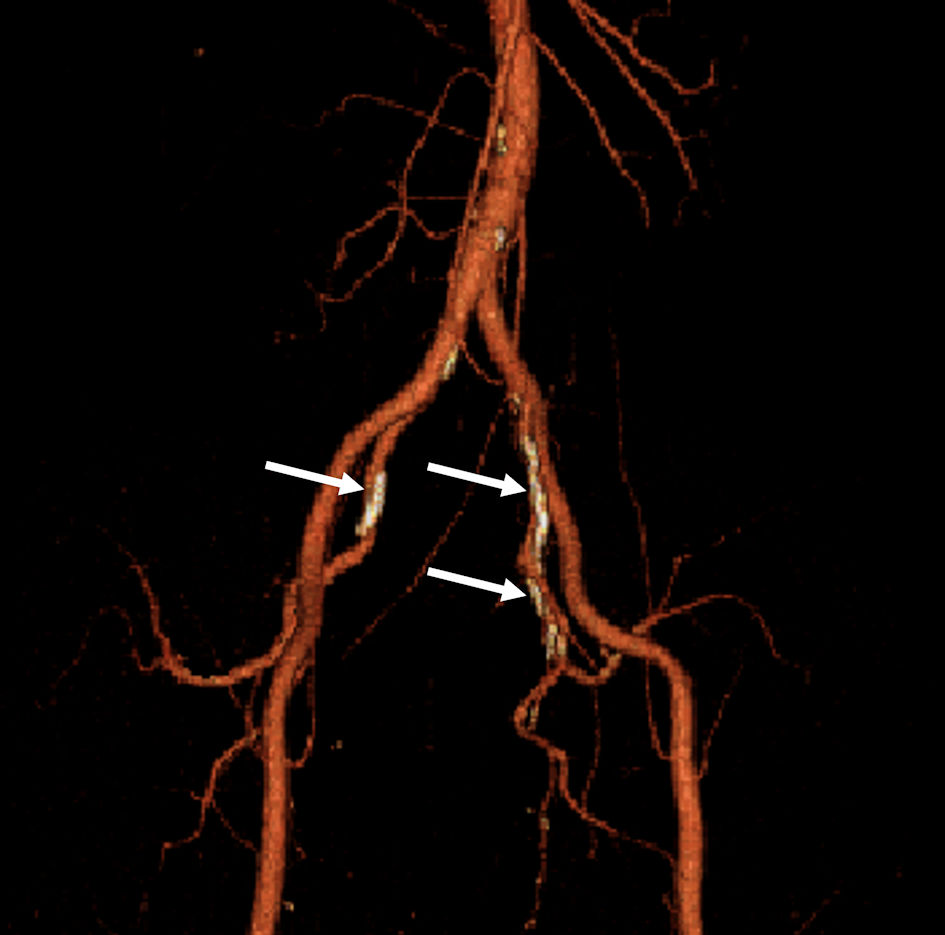 Click for large image |
Figure 4. Three-dimensional computed tomography (3DCT) of the abdominal aorta to arteria femoralis. Calcification was observed only in the internal iliac artery but not in the external iliac artery to the femoral artery (white arrows). |
Moreover, the coronary artery calcification score (CACS), which quantifies the calcification of the coronary arteries, was measured at the time of the second chest CT scan. The CACS was 214.0, indicating a moderate risk of coronary artery disease. However, this may be lower than in patients undergoing conventional dialysis.
The brachial-ankle pulse wave velocity (baPWV) indicates arterial stiffness and vascular calcification. The baPWV was measured pre-dialysis using an automated oscillometric device form (Nippon Colin, Japan). The baPWV 34 years after initiating hemodialysis was within the normal range (< 1,200), with right/left = 1,070/1,091 cm/s.
His nutritional status was assessed using two indices. The geriatric nutritional risk index (GNRI), which comprises albumin and body weight terms, has been reported as a simplified nutritional screening tool for maintenance hemodialysis patients. The cutoff value for malnutrition in the GNRI was < 91.2 [16]. Since the transfer, the GNRI has remained at 96.5 to 104.0, indicating adequate nutritional status. In addition, we evaluated the creatinine generation rate (CGR), which is an indicator of protein nutritional status and reflects muscle mass [17]. Higher values indicate more muscle mass, and values above 100% indicate above-average muscle mass. The rate was low at 92.0% at the time of transfer but increased to 111.0% 3 years later and then remained high, ranging from 109.0 to 126.6% (Fig. 1). The mean GNRI was 99.2 ± 4.0, and the mean CGR was 114.3±9.9 % (Table 1).
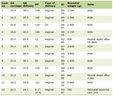 Click to view |
Table 1. Average Laboratory Data Over 27.5
Years After Transfer (December 1998 - May 2024) |
The calcium, phosphorus, and intact parathyroid hormone (iPTH) levels represent the average values at 1-year intervals from the time of transfer to our clinic. Serum calcium levels are shown as adjusted calcium values corrected for albumin. Throughout the study period, adjusted serum calcium levels averaged 8.8 ± 0.3 mg/dL, which was low but within the normal range. Serum phosphorus levels remained within normal limits, averaging 5.4 ± 0.5 mg/dL. The iPTH levels showed an upward trend after 17 years and remained somewhat high, ranging from 70.9 to 331.3 pg/mL. The mean calcium-phosphorus (Ca-P) product during the entire period was 47.9 ± 5.1. The phosphate binder used to lower serum phosphate levels was calcium carbonate. At the time of transfer, he was taking 5 g/day of calcium carbonate, but when he underwent extended-hours hemodialysis for 6 h, the dosage was reduced to 3 g/day. After 3.5 years, calcium carbonate was temporarily discontinued; however, a small dose of 1 g/day was reintroduced after 9 years. However, calcium carbonate was discontinued after 15.5 years when the dialysis duration was increased to 7 h. Calcium carbonate doses were low throughout the entire dialysis period. Active vitamin D3 was used to suppress iPTH levels. He was administered 2 µg/week of calcitriol at his transfer. After 7.5 years, it was temporarily discontinued; however, 11 years later, maxacalcitol 5 µg/week was started due to elevated iPTH levels. Maxacalcitol dosage was gradually increased to 10 µg/week to control iPTH levels (Fig. 1).
Laboratory data represent the mean (standard deviation (SD)) of the data obtained during the 27.5 years since the transfer to our clinic (Table 1). Serum phosphate levels, serum calcium, and iPTH levels, risk factors for vascular calcification, were 5.4 ± 0.5 mg/dL, 8.8 ± 0.3 mg/dL, and 197.8 ± 76.2 pg/mL, respectively. Albumin levels, an indicator of nutritional assessment, were well maintained at 4.0 ± 0.4 mg/dL. Triglyceride (TG), total cholesterol (TC), and low-density lipoprotein cholesterol (LDL-C) levels were slightly elevated.
Furthermore, the mean systolic and diastolic blood pressures during the 27.5 years of pre-dialysis were 118.5 ± 17.9 mm Hg and 73.9 ± 13.6 mm Hg, respectively, with a mean arterial pressure of 88.7 ± 15.0 mm Hg. He was normotensive and did not take any antihypertensive medications.
The dialysis conditions were as follows: the blood flow (Qb) was 178.8 ± 9.7 mL/min, and the dialysate flow (Qd) was 300 mL/min. The extended-hours hemodialysis without dietary restrictions treatment method is characterized by slower flow rates than conventional dialysis methods (Qb > 200 mL/min, Qd = 500 mL/min). However, the dialysis efficiency index single-pool KT/V (1.97 ± 0.2) and time-average concentration of urea (TAC-urea) (41.2 ± 7.2 mg/dL) showed sufficient values. The dialysate was Na+ = 140 mEq/L, K+ = 2.0 mEq/L, and Ca++ = 3.0 mEq/L (Table 2).
 Click to view |
Table 2. Average Hemodialysis Procedure Data
Over 27.5 Years After Transfer |
| Discussion | ▴Top |
In this case, the vascular calcification was slight despite being a long-term hemodialysis patient for 34 years. Vascular calcification is significantly more common in dialysis patients than in those without chronic kidney disease and contributes to the very high morbidity and mortality of cardiovascular disease [18].
We assessed vascular calcification in this case with AACI. Interestingly, despite a very long exposure to risk factors due to hemodialysis over 34 years, the AACI was low at 6.7%, with a slight 5-year change rate of +0.9%. In several reports, the average AACI of dialysis patients ranged from 22.1% to 80.2%, and the average duration of dialysis was less than 10 years [19-22]. Moriyama et al [21] reported AACI data for 94 patients, excluding those with diabetic nephropathy. The patients were divided into two groups: those with and those without cardiovascular disease. Among the 62 patients without cardiovascular disease comparable to the present case, with a mean age of 59.7 ± 1.4 years, the mean AACI was 34.2±3.4%. In contrast, the AACI in this case was 6.7%, approximately one-fifth of that observed in conventional dialysis.
However, the CACS, a measure of the risk of developing coronary artery disease, was moderate at 214.0. The CACS in dialysis patients has been reported to be significantly higher than that in the general population [23]. In addition, several reports indicate that the mean CACS of dialysis patients ranged from 449 to 4,290, and their mean duration of dialysis ranged from 5.3 to 10.4 years [24-29]. Also, to our knowledge, there are no studies on CACS in long-term dialysis patients beyond 11 years. They have also been reported to be male and have long-term hemodialysis vintage as independent risk factors for CAC progression [3]. Therefore, considering that he was a man who had been undergoing hemodialysis for 34 years, his CACS may have been lower than that of conventional dialysis patients.
Extended-hours hemodialysis has been reported to improve nutrition status and hypertension [10-13]. He has been undergoing 5-h dialysis since the initiation of hemodialysis. He underwent extended-hours hemodialysis without dietary restrictions 1.5 years after transfer to our clinic and continued to have 6 - 10 h of extended-hours hemodialysis sessions over the next 26 years. His mean systolic blood pressure measured before dialysis over the 27.5 years since his transfer to our clinic was 118 ± 17.9 mm Hg. He also maintained normal blood pressure without the use of antihypertensive medications. The severity of AACI reportedly increases with higher systolic blood pressure in dialysis patients [30].
Furthermore, the mean albumin level was 4.0 ± 0.4 mg/dL, and the mean GNRI, a nutritional index, was high at 99.2 ± 4.0, indicating adequate nutritional status over the 27.5 years since the transfer. Malnutrition in maintenance dialysis patients has been suggested to be an independent risk factor for aortic calcification progression, and improved nutrition may prevent aortic calcification [31].
Recently, CPPs have gained attention as risk factors for vascular calcification in dialysis patients. CPPs are blood-circulating colloidal particles formed from a combination of calcium phosphate and the serum protein fetuin-A [32]. CPPs are usually considered carriers of phosphate and calcium in the bone. These primary CPPs (CPP1) form small spherical colloidal nanoparticles that contain amorphous calcium phosphate. They prevent calcium and phosphate precipitation and are part of the blood mineral buffer system. However, in vitro, CPP1 aggregates and is converted into large, dense particles of secondary CPP (CPP2) containing crystalline calcium phosphate. The transition from amorphous calcium phosphate to crystalline calcium phosphate has been reported as a vascular calcification factor [33]. Blood CPP levels increase with CKD progression, and blood CPP2 levels are reportedly high in dialysis patients [34]. Nishibori et al [35] measured CPP2 in patients undergoing extended-hours hemodialysis without dietary restrictions at our clinic and compared it to the conventional hemodialysis group. Interestingly, blood CPP2 levels were significantly lower in the extended-hours hemodialysis without dietary restrictions group, at approximately half the level. Patients in the extended-hours hemodialysis group exhibited lower C-reactive protein (CRP) levels and higher serum albumin levels, suggesting a better nutritional and inflammatory status than those in the conventional hemodialysis group. Moreover, the findings imply that these patients may have higher fetuin-A levels. Based on these results, it has been reported that extended-hours hemodialysis without dietary restrictions may inhibit the formation of CPPs derived from serum phosphorus and calcium.
Several factors, including fetuin-A, pyrophosphate, magnesium, and zinc, inhibit vascular calcification [36]. Fetuin-A is a potent inhibitor of soft tissue calcification. Fetuin-A also stabilizes CPP1 and delays its progression to CPP2 [37]. Malnutrition decreases fetuin-A levels, which reduces vascular calcification. The GNRI has been reported to be an independent marker of fetuin-A levels [38]. The patient’s GNRI levels were high, indicating that his fetuin-A levels, which inhibit vascular calcification, may have been well maintained.
His serum cholesterol level was near the upper limit of normal. Dyslipidemia is considered a risk factor for causing calcification in atherosclerosis. However, there have been reports of an inverse correlation between serum cholesterol levels and risk for death in dialysis patients, known as the “cholesterol paradox”. It has also been reported that low cholesterol is associated with inflammation and nutritional disorders in dialysis patients [39, 40]. Evaluating chronic inflammation was unattainable because CRP was not a routine blood test in our clinic. However, the ferritin level, another indicator of chronic inflammation, was low at 59.0 ± 45.7 ng/mL [41]. Thus, he may have been well-nourished, and inflammation may have been suppressed. This relationship between malnutrition and inflammation is closely related to aortic calcification [7, 40].
Excessive calcium intake from calcium-based phosphate binders is associated with progressive vascular calcification [42]. In addition, higher serum calcium levels have been reported to increase the relative risk of death [43]. At the time of transfer, he was taking calcium carbonate 5 g/day, which he could reduce and discontinue because of the extended dialysis time. Albumin binds to ionized calcium (Ca2+) and traps Ca2+ in the blood [4]. His albumin levels were well maintained, and the serum calcium levels were near the low end of the normal range, at 8.8 ± 0.3 mg/dL. Calcium and phosphorus have been reported to synergize in promoting vascular calcification, with even a slight elevation in calcium levels significantly amplifying the pro-calcific effects of phosphorus [44]. His Ca-P product was normal at 47.9 ± 5.1. In addition, quantitative bone mineral density was assessed using calcaneal quantitative ultrasound (QUS) 34 years after the initiation of hemodialysis, at age 58 years. The speed of sound (SOS) was 1,608 m/s, the young adult mean (YAM) was 85.8%, and the T-score was -1.44, which was comparable to healthy individuals of the same age. These findings suggest that bone mineral metabolism may have been well controlled [45].
The uremic milieu is a significant risk factor for promoting vascular calcification, and not only calcium and phosphate load but also middle molecules (MMs), protein-bound uremic toxins (PBUTs), and low-molecular weight solutes uremic toxins were shown to affect vascular calcification [46-48]. Small and MMs are removed more adequately from the deeper compartments through extended-hours hemodialysis [49]. In addition, higher removal of PBUTs has been reported compared to conventional hemodialysis [50], as well as the biological improvement of vascular smooth muscle cells by extended-hours hemodialysis [51].
Calcification of the abdominal aorta was slight, but apparent calcification was observed in the internal iliac artery. Several possible reasons for this include the combined use of vitamin D3 and calcium carbonate in the early stages of induction and a history of smoking for several years at the time of hemodialysis initiation in his late 20s. However, to our knowledge, there are no reports of specific calcification of the internal iliac artery in dialysis patients. Continued follow-up is needed in the future.
General factors may be relevant to the slight vascular calcification, which was slight; he was 24 years old when he initiated hemodialysis, and he is still relatively young at 58 years old. Vascular calcification has been reported to be associated with aging [52], and its progression may be slower in younger individuals than in older individuals. However, vascular calcification has also been reported to be common and progressive in young adults on dialysis patients [6].
Extended-hours hemodialysis is efficacious in improving anemia, controlling hypertension, and removing phosphate [11-13]. It can also lead to reduced medication use and be beneficial to the economy. However, drawbacks include increased consumption of dialysate and electricity. Furthermore, when introducing extended-hours hemodialysis, it is essential to create an environment that supports this schedule, including the structure of the dialysis facility and staff cooperation with patients.
Despite these clinical advantages, extended treatment duration can impose psychological and quality-of-life burdens on patients, and obtaining informed consent remains a significant challenge. Nevertheless, nocturnal hemodialysis and home dialysis are viable modalities for extended-hours hemodialysis. Our clinic groups specialize in extended-hours hemodialysis without dietary restrictions and currently provide sessions exceeding 6 h to approximately 400 patients, both during the day and overnight.
Conclusions
This unique case is a testament to the potential benefits of extended-hours hemodialysis without dietary restrictions. The hemodialysis patients undergoing extended-hours hemodialysis without dietary restrictions may have well-controlled malnutrition, mineral metabolism, and a reduced risk of vascular calcification due to enhanced removal of uremic toxins. However, these findings are based on a single case, and further studies are necessary to extend these observations to a broader population.
Learning points
Increased vascular calcification is common in hemodialysis patients and has a significant negative impact on life prognosis. However, no standard treatment has been established at present. In this case, extended-hours hemodialysis without dietary restrictions was performed from the early stage of dialysis treatment, effectively slowing the progression of vascular calcification. Dialysis patients are exposed to many risk factors that promote vascular calcification, and it is important to eliminate them whenever possible. This treatment method significantly benefits dialysis patients by effectively removing uremic toxins, normalizing hypertension, and improving malnutrition. The risk factors could be removed by performing extended-hours hemodialysis without dietary restrictions, which may reduce vascular calcification.
Acknowledgments
The authors gratefully acknowledge Prof. Shoichi Maruyama, Dr. Takahiro Imaizumi and Dr. Bernard Charra for their kind comments.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
The patient gave consent to publish the data in this study.
Author Contributions
HK and TN contributed to the manuscript’s writing. KO, KK, YT, KS and FK reviewed and adjusted the manuscript before submission.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
CPPs: calciprotein particles; CT: computed tomography; AACI: abdominal aortic calcification index; CACS: coronary artery calcification score; baPWV: brachial-ankle pulse wave velocity; GNRI: geriatric nutritional risk index; CGR: creatinine generation rate; iPTH: intact parathyroid hormone; Ca-P product: calcium-phosphorus product; CPP1: primary CPPs; CPP2: secondary CPP; MMs: middle molecules; PBUTs: protein-bound uremic toxins
| References | ▴Top |
- Noordzij M, Cranenburg EM, Engelsman LF, Hermans MM, Boeschoten EW,
Brandenburg VM, Bos WJ, et al. Progression of aortic calcification is associated with disorders
of mineral metabolism and mortality in chronic dialysis patients. Nephrol Dial Transplant.
2011;26(5):1662-1669.
doi pubmed - Ohya M, Otani H, Kimura K, Saika Y, Fujii R, Yukawa S, Shigematsu T.
Vascular calcification estimated by aortic calcification area index is a significant predictive
parameter of cardiovascular mortality in hemodialysis patients. Clin Exp Nephrol.
2011;15(6):877-883.
doi pubmed - Siracusa C, Carabetta N, Morano MB, Manica M, Strangio A, Sabatino J,
Leo I, et al. Understanding vascular calcification in chronic kidney disease: pathogenesis and
therapeutic implications. Int J Mol Sci. 2024;25(23):13096.
doi pubmed - Heiss A, DuChesne A, Denecke B, Grotzinger J, Yamamoto K, Renne T,
Jahnen-Dechent W. Structural basis of calcification inhibition by alpha 2-HS
glycoprotein/fetuin-A. Formation of colloidal calciprotein particles. J Biol Chem.
2003;278(15):13333-13341.
doi pubmed - Viegas CSB, Santos L, Macedo AL, Matos AA, Silva AP, Neves PL, Staes
A, et al. Chronic kidney disease circulating calciprotein particles and extracellular vesicles
promote vascular calcification: a role for GRP (Gla-Rich Protein). Arterioscler Thromb Vasc
Biol. 2018;38(3):575-587.
doi pubmed - Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, Wang Y, et
al. Coronary-artery calcification in young adults with end-stage renal disease who are
undergoing dialysis. N Engl J Med. 2000;342(20):1478-1483.
doi pubmed - Choi SR, Lee YK, Cho AJ, Park HC, Han CH, Choi MJ, Koo JR, et al.
Malnutrition, inflammation, progression of vascular calcification and survival:
Inter-relationships in hemodialysis patients. PLoS One. 2019;14(5):e0216415.
doi pubmed - Lee SM, An WS. Supplementary nutrients for prevention of vascular
calcification in patients with chronic kidney disease. Korean J Intern Med.
2019;34(3):459-469.
doi pubmed - Shinaberger CS, Greenland S, Kopple JD, Van Wyck D, Mehrotra R,
Kovesdy CP, Kalantar-Zadeh K. Is controlling phosphorus by decreasing dietary protein intake
beneficial or harmful in persons with chronic kidney disease? Am J Clin Nutr.
2008;88(6):1511-1518.
doi pubmed - Kaneda H, Nishiyama T, Owada K, Katayose K, Takagi Y, Ishikawa S,
Tabata S, et al. Extended-hours hemodialysis without dietary restrictions increases body mass
index and normalizes hypertension: a case report. Tohoku J Exp Med.
2023;260(2):135-140.
doi pubmed - Demirci C, Ozkahya M, Demirci MS, Asci G, Kose T, Colak T, Duman S,
et al. Effects of three times weekly eight-hour nocturnal hemodialysis on volume and nutritional
status. Am J Nephrol. 2013;37(6):559-567.
doi pubmed - Raj DS, Charra B, Pierratos A, Work J. In search of ideal
hemodialysis: is prolonged frequent dialysis the answer? Am J Kidney Dis.
1999;34(4):597-610.
doi pubmed - Wong B, Collister D, Muneer M, Storie D, Courtney M, Lloyd A,
Campbell S, et al. In-center nocturnal hemodialysis versus conventional hemodialysis: a
systematic review of the evidence. Am J Kidney Dis. 2017;70(2):218-234.
doi pubmed - Yukawa S, Sonobe M, Tone Y, Yukawa A, Mimura K, Mune M, Maeda T, et
al. Prevention of aortic calcification in patients on hemodialysis by long-term administration
of vitamin E. J Nutr Sci Vitaminol (Tokyo). 1992;Spec No:187-190.
doi pubmed - Ohya M, Otani H, Kimura K, Saika Y, Fujii R, Yukawa S, Shigematsu T.
Improved assessment of aortic calcification in Japanese patients undergoing maintenance
hemodialysis. Intern Med. 2010;49(19):2071-2075.
doi pubmed - Yamada K, Furuya R, Takita T, Maruyama Y, Yamaguchi Y, Ohkawa S,
Kumagai H. Simplified nutritional screening tools for patients on maintenance hemodialysis.
Am J Clin Nutr. 2008;87(1):106-113.
doi pubmed - Shinzato T, Nakai S, Miwa M, Iwayama N, Takai I, Matsumoto Y, Morita
H, et al. New method to calculate creatinine generation rate using pre- and postdialysis
creatinine concentrations. Artif Organs. 1997;21(8):864-872.
doi pubmed - Disthabanchong S. Vascular calcification in chronic kidney disease:
Pathogenesis and clinical implication. World J Nephrol. 2012;1(2):43-53.
doi pubmed - Nitta K, Akiba T, Uchida K, Kawashima A, Yumura W, Kabaya T, Nihei H.
The progression of vascular calcification and serum osteoprotegerin levels in patients on
long-term hemodialysis. Am J Kidney Dis. 2003;42(2):303-309.
doi pubmed - Yamada K, Fujimoto S, Nishiura R, Komatsu H, Tatsumoto M, Sato Y,
Hara S, et al. Risk factors of the progression of abdominal aortic calcification in patients on
chronic haemodialysis. Nephrol Dial Transplant. 2007;22(7):2032-2037.
doi pubmed - Moriyama Y, Eriguchi R, Sato Y, Nakaya Y. Chronic hemodialysis
patients with visceral obesity have a higher risk for cardiovascular events. Asia
Pac J Clin Nutr. 2011;20(1):109-117.
pubmed - Yoon HE, Chung S, Whang HC, Shin YR, Hwang HS, Chung HW, Park CW, et
al. Abdominal aortic calcification is associated with diastolic dysfunction, mortality, and
nonfatal cardiovascular events in maintenance hemodialysis patients. J Korean Med Sci.
2012;27(8):870-875.
doi pubmed - Braun J, Oldendorf M, Moshage W, Heidler R, Zeitler E, Luft FC.
Electron beam computed tomography in the evaluation of cardiac calcification in chronic dialysis
patients. Am J Kidney Dis. 1996;27(3):394-401.
doi pubmed - Nitta K, Akiba T, Suzuki K, Uchida K, Ogawa T, Majima K, Watanabe R,
et al. Assessment of coronary artery calcification in hemodialysis patients using multi-detector
spiral CT scan. Hypertens Res. 2004;27(8):527-533.
doi pubmed - Tamashiro M, Iseki K, Sunagawa O, Inoue T, Higa S, Afuso H, Fukiyama K. Significant association between the progression of coronary artery calcification and dyslipidemia in patients on chronic hemodialysis. Clin Chim Acta. 2006;36:98-102.
- Ohtake T, Kobayashi S, Oka M, Furuya R, Iwagami M, Tsutsumi D,
Mochida Y, et al. Lanthanum carbonate delays progression of coronary artery calcification
compared with calcium-based phosphate binders in patients on hemodialysis: a pilot study.
J Cardiovasc Pharmacol Ther. 2013;18(5):439-446.
doi pubmed - Stompor T. Coronary artery calcification in chronic kidney disease:
An update. World J Cardiol. 2014;6(4):115-129.
doi pubmed - Gaubeta S, Klinghammer L, Jahn D, Schuhback A, Achenbach S, Marwan M.
Epicardial fat and coronary artery calcification in patients on long-term hemodialysis.
J Comput Assist Tomogr. 2014;38(5):768-772.
doi pubmed - Matsuura T, Abe T, Onoda M, Ikarashi D, Sugimura J, Komaki T, Sasaki
N, et al. Pelvic artery calcification score is a marker of vascular calcification in male
hemodialysis patients. Ther Apher Dial. 2018;22(5):509-513.
doi pubmed - Kimura K, Saika Y, Otani H, Fujii R, Mune M, Yukawa S. Factors
associated with calcification of the abdominal aorta in hemodialysis patients. Kidney Int Suppl.
1999;71:S238-241.
doi pubmed - Okamoto T, Tsutaya C, Hatakeyama S, Konishi S, Okita K, Tanaka Y,
Imanishi K, et al. Low serum butyrylcholinesterase is independently related to low fetuin-A in
patients on hemodialysis: a cross-sectional study. Int Urol Nephrol.
2018;50(9):1713-1720.
doi pubmed - Kutikhin AG, Feenstra L, Kostyunin AE, Yuzhalin AE, Hillebrands JL,
Krenning G. Calciprotein particles: balancing mineral homeostasis and vascular pathology.
Arterioscler Thromb Vasc Biol. 2021;41(5):1607-1624.
doi pubmed - Kuro OM. Calcium phosphate microcrystallopathy as a paradigm of
chronic kidney disease progression. Curr Opin Nephrol Hypertens. 2023;32(4):344-351.
doi pubmed - Hamano T, Matsui I, Mikami S, Tomida K, Fujii N, Imai E, Rakugi H, et
al. Fetuin-mineral complex reflects extraosseous calcification stress in CKD. J Am Soc
Nephrol. 2010;21(11):1998-2007.
doi pubmed - Nishibori N, Okazaki M, Miura Y, Hishida M, Kurasawa S, Imaizumi T,
Kato N, et al. Association of calciprotein particles with serum phosphorus among patients
undergoing conventional and extended-hours haemodialysis. Clin Kidney J.
2024;17(6):sfae121.
doi pubmed - Heiss A, Eckert T, Aretz A, Richtering W, van Dorp W, Schafer C,
Jahnen-Dechent W. Hierarchical role of fetuin-A and acidic serum proteins in the formation and
stabilization of calcium phosphate particles. J Biol Chem.
2008;283(21):14815-14825.
doi pubmed - Schafer C, Heiss A, Schwarz A, Westenfeld R, Ketteler M, Floege J,
Muller-Esterl W, et al. The serum protein alpha 2-Heremans-Schmid glycoprotein/fetuin-A is a
systemically acting inhibitor of ectopic calcification. J Clin Invest.
2003;112(3):357-366.
doi pubmed - Okamoto T, Hatakeyama S, Kodama H, Horiguchi H, Kubota Y, Kido K,
Momota M, et al. The relationship between poor nutritional status and progression of aortic
calcification in patients on maintenance hemodialysis. BMC Nephrol. 2018;19(1):71.
doi pubmed - Liu Y, Coresh J, Eustace JA, Longenecker JC, Jaar B, Fink NE, Tracy
RP, et al. Association between cholesterol level and mortality in dialysis patients: role of
inflammation and malnutrition. JAMA. 2004;291(4):451-459.
doi pubmed - Kalantar-Zadeh K, Block G, Humphreys MH, Kopple JD. Reverse
epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney Int.
2003;63(3):793-808.
doi pubmed - Maruyama Y, Yokoyama K, Yokoo T, Shigematsu T, Iseki K, Tsubakihara
Y. The different association between serum ferritin and mortality in hemodialysis and peritoneal
dialysis patients using Japanese nationwide dialysis registry. PLoS One.
2015;10(11):e0143430.
doi pubmed - Guerin AP, London GM, Marchais SJ, Metivier F. Arterial stiffening
and vascular calcifications in end-stage renal disease. Nephrol Dial Transplant.
2000;15(7):1014-1021.
doi pubmed - Block GA, Klassen PS, Lazarus JM, Ofsthun N, Lowrie EG, Chertow GM.
Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc
Nephrol. 2004;15(8):2208-2218.
doi pubmed - Reynolds JL, Joannides AJ, Skepper JN, McNair R, Schurgers LJ,
Proudfoot D, Jahnen-Dechent W, et al. Human vascular smooth muscle cells undergo
vesicle-mediated calcification in response to changes in extracellular calcium and phosphate
concentrations: a potential mechanism for accelerated vascular calcification in ESRD.
J Am Soc Nephrol. 2004;15(11):2857-2867.
doi pubmed - Toussaint N, Boddington J, Simmonds R, Waldron C, Somerville C, Agar
J. Calcium phosphate metabolism and bone mineral density with nocturnal hemodialysis. Hemodial
Int. 2006;10(3):280-286.
doi pubmed - Nikolov I, Joki N, Drueke T, Massy Z. Beyond phosphate—role of
uraemic toxins in cardiovascular calcification. Nephrol Dial Transplant.
2006;21(12):3354-3357.
doi pubmed - Rapp N, Evenepoel P, Stenvinkel P, Schurgers L. Uremic toxins and
vascular calcification-missing the forest for all the trees. Toxins (Basel).
2020;12(10):624.
doi pubmed - Chao CT, Lin SH. Uremic vascular calcification: the pathogenic roles
and gastrointestinal decontamination of uremic toxins. Toxins (Basel). 2020;12(12):812.
doi pubmed - Basile C, Libutti P, Di Turo AL, Casino FG, Vernaglione L, Tundo S,
Maselli P, et al. Removal of uraemic retention solutes in standard bicarbonate haemodialysis and
long-hour slow-flow bicarbonate haemodialysis. Nephrol Dial Transplant.
2011;26(4):1296-1303.
doi pubmed - Cornelis T, Eloot S, Vanholder R, Glorieux G, van der Sande FM,
Scheijen JL, Leunissen KM, et al. Protein-bound uraemic toxins, dicarbonyl stress and advanced
glycation end products in conventional and extended haemodialysis and haemodiafiltration.
Nephrol Dial Transplant. 2015;30(8):1395-1402.
doi pubmed - Chan CT, Lovren F, Pan Y, Verma S. Nocturnal haemodialysis is associated with improved vascular smooth muscle cell biology. Nephrol Dial Transplant. 2009;24:2915-2919.
- Witteman JC, Kannel WB, Wolf PA, Grobbee DE, Hofman A, D'Agostino RB,
Cobb JC. Aortic calcified plaques and cardiovascular disease (the Framingham Study).
Am J Cardiol. 1990;66(15):1060-1064.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Medical Cases is published by Elmer Press Inc.