| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://jmc.elmerpub.com |
Case Report
Volume 16, Number 3, March 2025, pages 107-113
Successful Sentinel Lymph Node Biopsy in Accessory Breast Cancer
Young Duck Shina , Young Jin Choib, c
aDepartment of Anesthesiology, Chungbuk National University Hospital, Chungbuk
National University College of Medicine, Cheongju 28644, Korea
bDepartment of
Surgery, Chungbuk National University Hospital, Chungbuk National University College of
Medicine, Cheongju 28644, Korea
cCorresponding Author: Young Jin Choi,
Department of Surgery, Chungbuk National University Hospital, Chungbuk National University
College of Medicine, Seowon-gu, Cheongju 28644, Korea
Manuscript submitted December 10, 2024, accepted January 1, 2025, published online February 18,
2025
Short title: Sentinel Node Biopsy in Accessory Breast Cancer
doi:
https://doi.org/10.14740/jmc5094
| Abstract | ▴Top |
Primary breast cancer occurring in accessory breast tissue is exceptionally rare, with an incidence of 0.2-0.6%. It can aggressively progress, often leading to early metastasis. Treatment is typically delayed due to the rarity, variety of differentials, and lack of clinical awareness of the disease. In axillary surgery, sentinel lymph node mapping in patients with axillary breast cancer is technically challenging and has been poorly described. Here, we present a case of a 53-year-old woman with a 0.5 × 1 cm hard lump in the right axillary region for 2 years, progressive growth for 6 months, and no concomitant breast lesion or axillary lymphadenopathy. Core needle biopsy revealed invasive ductal carcinoma with estrogen receptor and progesterone receptor expression and human epidermal growth factor receptor 2 negativity, whereas mammography and breast magnetic resonance imaging revealed no primary breast lesions. She was diagnosed with invasive cancer arising from an accessory breast and underwent wide total excision of the right accessory breast and sentinel lymph node biopsy. Sentinel lymph node biopsy can be successfully performed using intratumoral dye and subareolar radiocolloid mapping in accessory breast cancer surgery. Axillary accessory breast tissue is outside the scope of the screening breast examination; therefore, oncologists must be aware of this entity and associated pathologies.
Keywords: Accessory breast; Accessory breast carcinoma; Sentinel lymph node biopsy; Sentinel lymph node mapping; Radiotherapy
| Introduction | ▴Top |
Primary breast cancer occurring in accessory breast tissue is very rare, with an incidence of 0.2-0.6%. It may follow an aggressive course with a tendency toward early metastasis. Owing to its rarity, variety of differentials, and lack of clinical awareness, treatment is usually delayed [1]. The proximity of the primary tumor to the axillary lymph nodes makes axillary management in patients with accessory breast cancer challenging. Routine axillary dissection has been recommended by surgeons as a part of primary surgery due to the higher rate of nodal involvement and likelihood of obscuration of sentinel nodes following tracer injection, given their proximity to the tumor [2]. Sentinel lymph node mapping in patients with axillary breast cancer is technically challenging and remains poorly described [2, 3]. Because the embryological development of the accessory breast occurs independently from that of the breast, its lymphatic drainage may likely differ. Whether the tracer injection should be located peritumorally in the axillary or periareolar region of the breast remains to be determined [4].
Accessory breast cancer, also known as ectopic breast cancer, refers to the development of malignant tumors in the accessory breast tissue, which arises from remnants of mammary gland tissue that fail to regress during fetal development. Accessory breast tissue can occur along the “milk line,” which extends from the axilla to the groin and undergoes hormonal changes similar to normal breast tissue; therefore, accessory breasts are susceptible to both benign and malignant conditions, even outside the usual breast area [4]. The clinical presentation of accessory breast cancer significantly varies based on its atypical location and may be discovered incidentally during imaging studies or surgical procedures for unrelated reasons. Although rare, this condition poses significant clinical challenges and requires a nuanced understanding of its presentation and management [1, 2].
Here, we present a case of a 53-year-old woman with a hard lump in the right axillary region for 2 years, progressive growth for 6 months, and no concomitant breast lesion or axillary lymphadenopathy.
| Case Report | ▴Top |
Investigations
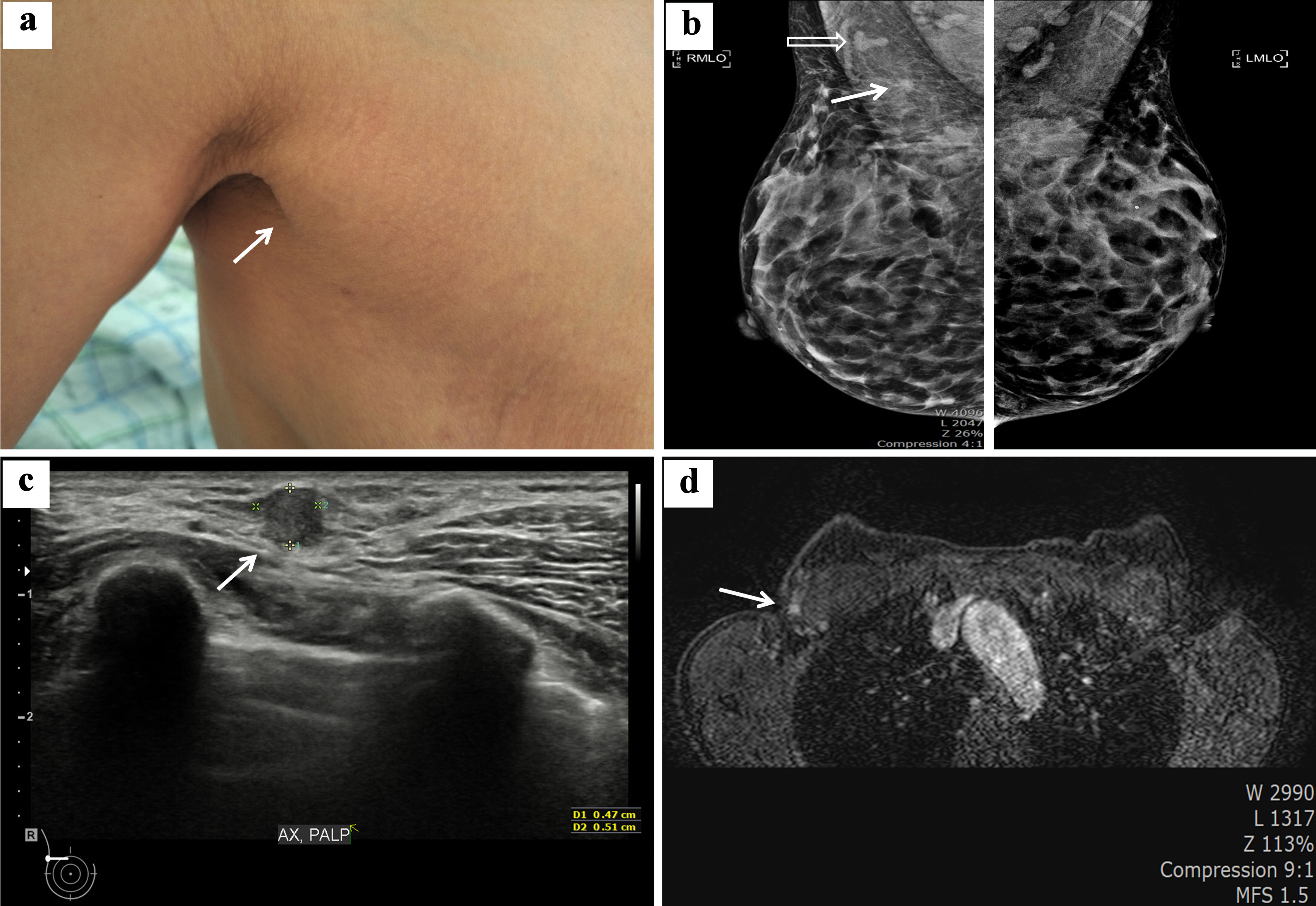
A 53-year-old woman presented to our breast clinic with a palpable axillary mass. She indicated that the mass had been detected 2 years prior and had progressively grown over the last 6 months. She also denied a family history of breast or ovarian cancer. Upon physical examination, a slightly movable hard mass (0.5 × 1 cm) was palpated on the right axilla, with no evidence of abnormality in the bilateral breasts. Accessory breasts were found bilaterally in the axilla, and a nodular lesion was suspected in the right accessory breast. The patient had a thin body habitus, and the right axillary mass was in the subcutaneous skin (Fig. 1a).
 Click for large image |
Figure 1. Clinical and radiologic findings of the right axilla mass diagnosed as invasive tubular carcinoma. (a) Exposed axilla shows right axillary swelling compatible with accessory breast (white arrow). (b) Mammographic image in the mediolateral oblique position revealing bilateral axillary accessory breast and demonstrating a 0.6 cm irregular asymmetric density (white arrow) and a suspicious lymph node (white nonshaded arrow) in the right axilla. (c) Breast ultrasonographic imaging shows a 0.51 × 0.47 cm irregular shaped hypoechoic mass located in the right axilla (white arrow). (d) Contrast- enhanced T1 breast magnetic resonance imaging shows a 0.6 cm, ill-defined enhancing nodule on the right axilla (white arrow). |
Diagnosis
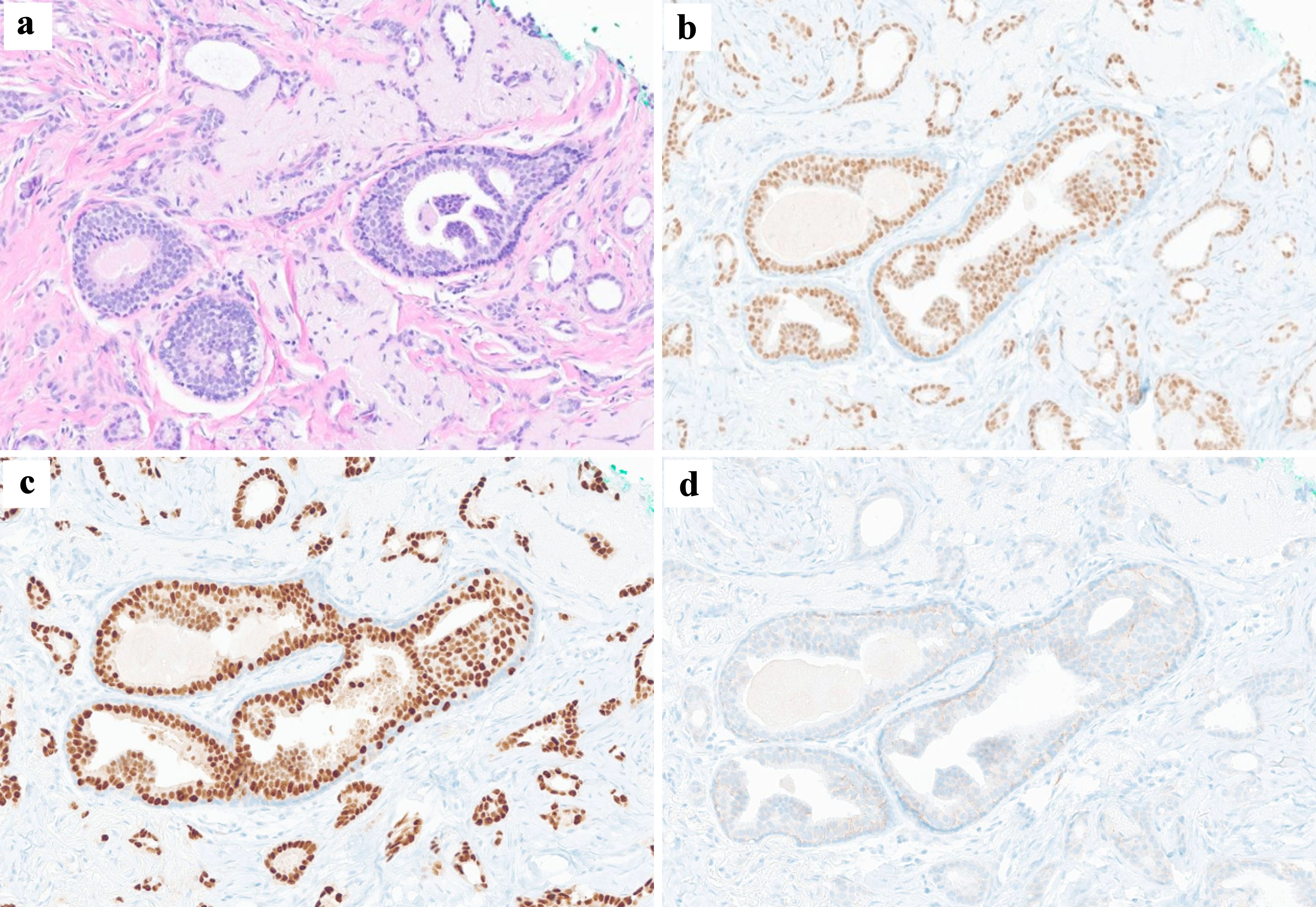
Mammography showed that the breasts were heterogeneously dense, with no suspicious findings suggesting malignancy in either breast (Fig. 1b). Breast ultrasonography (USG) showed accessory breast in bilateral axilla and irregular heterogenous hypoechoic mass (0.51 × 0.47 cm in size) with nonparallel orientation in right axillary breast tissue (Fig. 1c). The lymph nodes in the axillary fossa were normal in size and shape. USG-guided core needle biopsy of the right axillary mass revealed low-grade invasive ductal carcinoma with estrogen receptor (ER) and progesterone receptor (PR) expression and negative human epidermal growth factor receptor 2 (HER2) expression (Fig. 2a-d).
 Click for large image |
Figure 2. Histologic findings of core needle biopsy specimen. (a) Histopathology shows low-grade invasive ductal carcinoma with tubular pattern, with no marked pleomorphism of the nuclei (hematoxylin and eosin staining × 100). The primary breast carcinoma was immunohistochemically positive for estrogen receptor (b) and progesterone receptor (c) and negative for C-erb-B2 (d). |
The axillary lesion displays pathological features of invasive carcinoma, characterized by well-formed tubules within the benign breast and adipose tissue, confirming the presence of invasive cancer originating from the axillary accessory breast. Breast magnetic resonance imaging (MRI) revealed no evidence of a primary breast lesion. T1 fat-saturated postcontrast sagittal and coronal breast MRI demonstrated an irregularly enhancing mass in the right axilla (Fig. 1d). Chest and abdominal computed tomography (CT) and whole-body bone scans revealed the absence of distant metastases.
Treatment
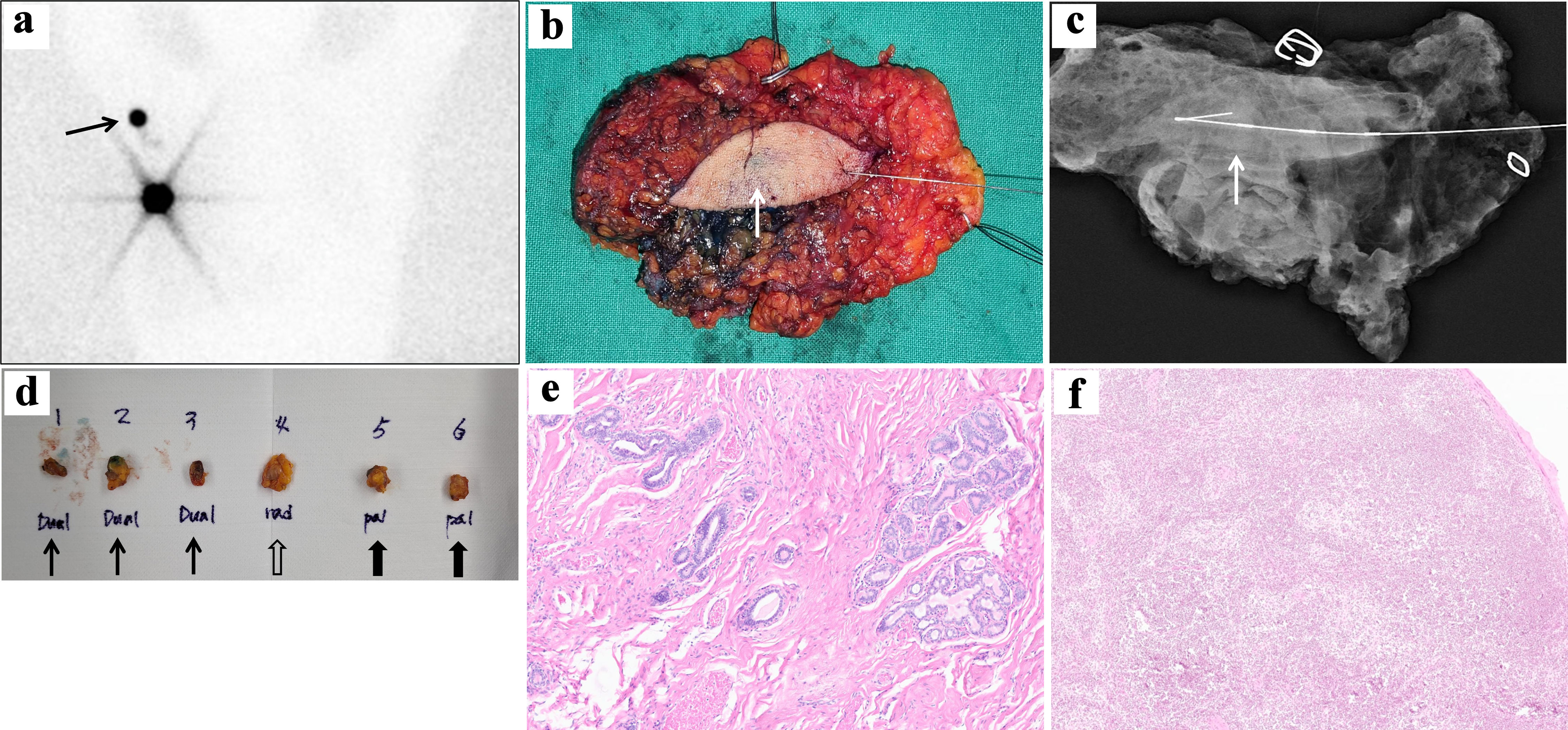
After the diagnosis of invasive cancer arising from an accessory breast, she underwent wide total excision of the right accessory breast, and sentinel lymph node biopsy was performed. Twelve hours prior to surgery, 400 µCi of technetium-99m sulfur colloid was injected into the right subareolar region, and lymphoscintigraphy was obtained 10 and 60 min after the study (Fig. 3a). After completing general anesthesia, 5 mL of indigo-carmine solution was injected directly into the axillary mass under breast USG guidance. An elliptical incision was made over the mass and accessory breast, and deepened using cautery. The whole accessory breast tissue, including the tumor, was resected along with an ellipse of the overlying skin and a rim of normal adipose tissue. The posterior border of the tumor consisted of the pectoralis major muscle, the fascia of which was included in the primary specimen (Fig. 3b, c). Additional shaved margins were obtained, and a frozen biopsy of the cavitary margin in eight directions revealed no evidence of malignancy.
 Click for large image |
Figure 3. Operative procedures and findings of the patient. (a) Sixty minutes delayed image of preoperative lymphoscintigraphy after subareolar injection of 400 µCi technetium-99m sulfur colloid clearly visualizes right axillary sentinel lymph node (black arrow). (b) After intraoperative ultrasonography-guided wire localization of axilla mass, total excision of right axillary accessory breast including malignancy was performed. The intratumoral blue indigo-carmine injection is identified (white arrow). (c) Specimen mammography shows centrally located wire and poorly defined asymmetric density (white arrow). (d) Three dual tracer-uptake sentinel lymph nodes (black arrow) and one radioactive sentinel lymph node (black nonshaded arrow) were dissected through dual mapping with a subareolar radionuclide tracer and intratumoral indigo-carmine injections. Two additional palpable sentinel lymph nodes (black shaded arrow) were also dissected. (e) Histologic finding of breast carcinoma on breast conserving surgery (BCS) specimen showed low-grade invasive tubular carcinoma, with tubular growth in > 90% of tumor, with ovoid tubules with open lumina, with no marked pleomorphism of the nuclei and no increased mitotic activity (hematoxylin and eosin staining × 40). (f) Sentinel lymph node showed no evidence of metastasis (hematoxylin and eosin staining × 40). |
Three dual tracer-uptake sentinel lymph nodes and one radioactive sentinel lymph node were dissected through dual mapping with a subareolar radionuclide tracer and intratumoral indigo-carmine injections. Two additional palpable sentinel lymph nodes were dissected; none of the six sentinel lymph nodes were involved in the frozen biopsy (Fig. 3d). The entire axilla was rescanned, and no other areas of tracer uptake were identified. No additional suspicious lymph nodes were found upon palpation or inspection.
A diagnosis of invasive tubular carcinoma (0.7 × 0.6 cm in size) in the accessory breast, with no evidence of sentinel lymph node metastasis, was made (Fig. 3e, f). Tumor revealed low nuclear grade and low histologic grade, with ER (2+, > 95%) and PR expression (3+, > 95%), negative HER2 expression, and a low Ki 67 proliferation index rate (2%). The six sentinel lymph nodes sampled were also negative for malignancy on permanent pathology. The surgical margins were negative, and according to the eighth edition of the American Joint Committee on Cancer staging system, the pathological stage of the tumor was stage I.
Two days after surgery, the patient was discharged without any complications. Adjuvant endocrine therapy with an aromatase inhibitor (anastrozole, 1 mg/day) was initiated, and she underwent adjuvant radiation therapy at 5,000 cGy on her right breast and axilla.
Follow-up and outcomes
Currently, 26 months after surgery, the patient continues endocrine therapy with anastrozole, and no evidence of recurrence has been observed in breast MRI.
| Discussion | ▴Top |
Herein, we report a case of atypical breast cancer in a 53-year-old woman with a progressive palpable axillary mass. Initial imaging findings of the primary clinical ultrasound suggested the presence of benign lymphadenopathy; however, a subsequent core needle biopsy indicated an invasive ductal carcinoma of the axillary accessory breast. This case highlights the importance of considering accessory breast cancers in the differential diagnosis of atypical axillary masses.
Diagnosis of accessory breast cancer necessitates thorough clinical examination and imaging techniques, including USG, mammography, and MRI [5, 6]. Accurate imaging is vital for differentiating between benign and malignant processes in the axillary breast tissue. USG is often the first-line noninvasive imaging modality for evaluating palpable masses in the axillary region. It can help differentiate between solid masses and cystic lesions in the axilla and other possible axillary masses, including lymph nodes. However, this procedure is operator-dependent and may not provide comprehensive information on deeper structures. Mammography is also recommended for screening and assessing breast tissue in conventional and accessory breast regions. It can effectively identify microcalcifications and solid masses. However, it may have limited sensitivity for visualizing lesions in the axillary area due to overlapping structures. In contrast, MRI can be used to evaluate suspicious findings from ultrasound or mammography, especially in high-risk patients and those with excellent soft tissue contrast. MRI is highly sensitive in detecting breast cancer, including axillary tissue [5, 6]; however, it is costly and time-consuming, compared with other modalities, and may require the use of contrast agents. In our patient, the initial ultrasound evaluation revealed a solid lesion with suspicious characteristics, irregular margins, and increased vascularity. Initial ultrasound imaging suggested atypical lymphadenopathy with cortical thickening; however, a subsequent mammogram showed ambiguous accessory breast tissue in the corresponding area, highlighting the necessity for a targeted biopsy.
Accessory breast cancer and axillary tail breast cancer are rare forms of breast cancer arising from ectopic breast tissue, which may lead to diagnostic challenges and significantly impact clinical outcomes and management strategies. Axillary tail breast cancer refers specifically to malignancies that arise from the axillary tail of the breast, an extension of the breast tissue into the axilla, and often presents with clinical features similar to those of accessory breast cancer [7]. Owing to its high sensitivity and superior soft-tissue contrast, MRI provides excellent contrast between normal and abnormal tissues, allowing for clear visualization of tumors within the accessory breast tissue or the axillary tail [5, 6]. In addition, the ability to view images in multiple planes aids in accurately locating lesions, which is essential for surgical planning [6]. Accessory breast cancer can arise in the subcutaneous layer of the axillae, showing discontinuity with the normal pectoral breast and leading to a potential misdiagnosis as a skin lesion. In contrast, axillary tail breast cancer can be obscured by the pectoralis muscle or plastic reconstruction, leading to its potential detection at a later stage [7]. Axillary tail breast cancer can have a similar enhancement to accessory breast cancer; however, the pattern may differ based on the proximity of the tumor to lymphatic structures, potentially displaying a more heterogeneous enhancement and typically showing more pronounced inflammatory changes or lymphadenopathy in the adjacent axillary lymph nodes, indicating potential metastasis [5-7]. In this case, MRI confirmed accessory breast cancer as the presence of a 0.6-cm irregular mass in the subcutaneous layer of the axillae, with significant enhancement as an isolated lesion in the vicinity of additional breast tissue and suspicious lymphadenopathy in the deep axilla.
Given its rarity and potential for misdiagnosis, a high index of suspicion and thorough evaluation are necessary when addressing palpable masses in accessory breast tissues [1, 2, 8]. A definitive diagnosis can only be made through the histological evaluation of biopsy samples, which can allow for timely and appropriate treatment. Core biopsy can provide a core of tissue, allowing for better architecture preservation and accurate tissue evaluation, including immunohistochemical staining. In our patient, a subsequent USG-guided core needle biopsy confirmed the diagnosis of low-grade invasive ductal carcinoma in the right accessory breast tissue with ER and PR expression and negative HER2 expression.
Surgical treatment for accessory breast cancer must be personalized based on tumor characteristics, patient health, patient preferences, and the desire for breast preservation versus comprehensive treatment [8-10]. However, the first step of breast surgery is generally a wide local excision involving the removal of cancerous tissue along with a healthy clear margin. Some scholars believe that radical mastectomy is feasible in the presence of regional lymph node infiltration. However, Evans et al found that radical mastectomy or modified radical mastectomy had no obvious advantage in prognosis, compared with local resection plus axillary dissection or radiotherapy [1, 10, 11]. Additionally, Cogswell et al [12] reported that ipsilateral mastectomy had no significant effect on the prognosis of patients with ectopic breast cancer. Evans et al reviewed 90 cases of ectopic breast tissue carcinoma and found no survival advantage for patients undergoing radical or modified radical mastectomy over those undergoing local excision combined with axillary dissection or radiation [1]. Although no formal guidelines exist, ipsilateral mastectomy is not recommended unless there is an additional breast lesion. If clinical examinations and investigations, such as mammography and MRI, exclude its presence, the patient can be spared mastectomy with a close follow-up. Therefore, local wide excision with clear resection margin combined with axillary lymph node dissection, followed by radiotherapy, chemotherapy, and endocrine therapy is a viable treatment option [1, 10-12].
Due to their close proximity, axillary metastases have been reported at presentation in approximately 50% of accessory breast cancer patients [1, 2, 8]. Management of the axillary lymph nodes is sometimes difficult as the location of the tumor makes dissection challenging, especially in cases with severe adhesion or local invasion of the lymph nodes by the tumor. Accordingly, many surgeons recommend routine axillary dissection as part of primary surgery due to the higher rate of nodal involvement and potential for obscuration of sentinel nodes following tracer injection, given their proximity to the tumor [8, 10, 13]. Recent research indicates that accessory breast cancer can exhibit similar patterns of lymphatic spread as conventional breast cancer, with sentinel lymph node involvement in 20-30% of cases [2, 3, 13]. Early-stage accessory breast cancer treated with appropriate surgical interventions, including sentinel lymph node dissection (SLND), has shown favorable survival outcomes comparable to those of patients with typical breast cancer. However, sentinel lymph node mapping in patients with axillary breast cancer is technically challenging and poorly understood. Preoperative lymphoscintigraphy can be used for better localization of sentinel nodes. Localization of the tracer injection remains debatable; most reports favor peritumoral injection, although it can make detection of lymph nodes by gamma probes difficult due to the high background concentration of the tracer. The proximity of the axillary tumor to the regional lymph nodes may lead to a “shine-through” effect, by which it becomes difficult to identify an area of tracer localization in the axilla as a result of the lower gamma counts from the node(s) being obscured by the higher counts at the injection site. Accordingly, Harris et al recommend radiotracer injection after tumor excision [3]. In our patient, we used dual lymphatic mapping with a preoperative injection of subareolar radiocolloid and intratumoral indigo carmine vital dye.
After wide excision of the accessory breast cancer, successful sentinel lymph node detection can be performed to detect three dual tracer-taken sentinel lymph nodes and one radioactive sentinel lymph node. There was no evidence of blue dye spillage or excessive background radioactivity. Dual mapping with different localization sites may be a reasonable sentinel lymph node mapping method for accessory breast cancer surgeries. SLND plays a vital role in staging and therapeutic decisions, allowing for accurate assessment, while minimizing surgical morbidity. Axillary lymph node dissection should be performed only if sentinel lymph node metastasis is present.
The literature offers no formal guidance on adjuvant radiotherapy or its dose, fractionation, or the extent of treatment for the ipsilateral breast. In most case reports, radiotherapy has been recommended to maximize locoregional control. In a 2009 review of case reports, Madej et al [14] reported that most radiation prescriptions were 5,000 - 6,600 cGy at 200 cGy per fraction. More recently, Hallam et al [15] proposed an extrapolation from the UK START B trial, in which 4,000 cGy was administered over 15 fractions. Notably, there is no consensus on the treatment fields. The proposed plans range from treating the tumor bed alone to including the ipsilateral uninvolved pectoral breast, axilla, and supraclavicular fossa, even in the absence of nodal metastases. This lack of consensus is possibly attributed to differences in the classification of the extent of surgery. Considering that accessory breast tissue is an independent structure away from the pectoral breast tissue, this surgery may be defined as mastectomy if all accessory breast tissue including cancer was removed. If so, one decision-making approach would be to extrapolate the indications for postmastectomy radiation therapy from classic studies, that is, only offer it in cases of nodal involvement, T3 or T4 disease, and positive margins. If, on the other hand, this is considered a lumpectomy rather than a mastectomy in a low-risk patient, one may consider applying criteria from the Hughes et al trial (CALGB C9343) [16] for patients aged > 70 years, or the PRIME II trial [17] to identify those in whom postoperative radiation therapy is omitted. Additionally, if radiation is offered as part of breast conservation therapy, another potential option is accelerated partial breast irradiation. Accordingly, in a small series of case reports, the indications for radiotherapy were appropriately relaxed and tailored according to the patient’s risk of recurrence, stage, and nodal status [18]. Considering the lack of evidence assessing the middle- and long-term efficacy of radiation therapy to the tumor bed and ipsilateral breast, our patient underwent adjuvant radiation therapy with 5,000 cGy on her whole right breast and axilla after a multidisciplinary meeting.
Similar to conventional breast cancers, systemic therapy plays a crucial role in the overall management of accessory breast cancers, particularly in cases with invasive characteristics or metastasis. Evidence suggests that patients with accessory breast cancer treated with systemic therapies have survival rates comparable to those of patients with standard breast cancer, especially when tailored therapies are used [1, 2, 8-10]. Systemic adjuvant or neoadjuvant chemotherapy, such as anthracyclines or taxanes, is administered to patients with accessory breast cancer based on the same principles as those for primary breast cancer. Endocrine therapy should be administered to patients with receptor-positive disease. Because our patient had a low-grade endocrine-responsive stage I breast cancer, she did not undergo adjuvant chemotherapy, and adjuvant endocrine therapy with an aromatase inhibitor was initiated. The management of accessory breast cancers follows the same principles as pectoral breast cancer guidelines and is largely based on tumor subtype and stage [1, 2, 8-10]. But there are several issues that are essentially different from pectoral breast cancer and require a unique approach. Although rare entity breast cancer, larger studies are necessary to validate these findings.
The prognosis of accessory breast carcinoma is challenging to establish, primarily due to the absence or limited follow-up data, as well as small sample sizes. Additionally, it is challenging to identify clear histopathological and clinical distinctions between accessory and normal breast carcinomas. In a Japanese study [19], 68 cases of accessory breast cancer were followed up with a mean follow-up duration of 28.3 months (range, 2 - 156 months). The authors reported only one death due to pneumonia during the perioperative period. However, other studies have reported that the outcomes of ectopic breast cancer are poorer than those of normal breast cancer. This is primarily because the tumor is located near the axillary lymph nodes and, therefore, is associated with early metastasis to these nodes. The poorer outcomes in ectopic breast cancer relative to general breast cancer reflect less effective clinical disease management, compared with the disease process itself [20].
Follow-up data are provided for a limited number of patients, indicating a 4-year post-treatment survival of 9.4%, calculated in a meta-analysis [1]. Although no long-term follow-up data regarding the prognosis of accessory breast cancer are available, recent clinical awareness and advanced diagnostic imaging and screening techniques can lead to early diagnosis of accessory breast cancer. Patients with accessory breast cancer might not have a worse prognosis, compared with those with conventional breast cancer in the same stage. Twenty-six months after surgery, our patient was still undergoing endocrine therapy with anastrozole, and no evidence of recurrence was observed.
This case report described a rare breast carcinoma that arose in the accessory breast tissue. As accessory breast tissue is not detected on screening breast examinations, oncologists must be aware of this entity and associated pathologies. Correct diagnosis of disease extent and clear differentiation from axillary tail cancer can tailor adequate local therapy and prevent misdiagnosis and unnecessary surgery. Sentinel lymph node biopsies can be successfully performed using intratumoral dyes and subareolar radiocolloid mapping in accessory breast cancer surgery.
The management of accessory breast cancers follows the same principles as pectoral breast cancer guidelines and is largely based on tumor subtype and stage [1, 2, 8-10]. But there are several issues that are essentially different from pectoral breast cancer and require a unique approach. Although rare, larger studies are necessary to validate these findings.
Learning points
Accessory breast cancer presents a unique challenge in the field of oncology; and due to its rarity, there’s no formal guideline for tailored management strategies. Sentinel lymph node biopsy plays a vital role in staging and therapeutic decisions in early-stage accessory breast cancer patients. Dual mapping method with intra-tumoral dye and subareolar radiocolloid injection is an appropriate option for improving sentinel lymph node detection rate in accessory breast cancer patients.
Acknowledgments
None to declare.
Financial Disclosure
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from the patient for this case report.
Author Contributions
Shin YD and Choi YJ participated in the collection of medical data, wrote the manuscript and approved the final manuscript.
Data Availability
All data in our report were obtained from the patient’s hospitalization. Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
| References | ▴Top |
- Evans DM, Guyton DP. Carcinoma of the axillary breast.
J Surg Oncol. 1995;59(3):190-195.
doi pubmed - Sghaier S, M GH, Marghli I, Bouida A, Ben Hassouna J,
Chargui R, Rahal K. Primary ectopic axillary breast cancer: a case series. J Med Case
Rep. 2021;15(1):412.
doi pubmed - Harris MK, Guo MZ, Mangino A, Taylor C, Carson WE. Sentinel
node mapping and biopsy in ectopic axillary breast cancer: A case report and review of the
literature. Clin Case Rep. 2022;10(9):e6052.
doi pubmed - Velanovich V. Ectopic breast tissue, supernumerary breasts,
and supernumerary nipples. South Med J. 1995;88(9):903-906.
doi pubmed - DeFilippis EM, Arleo EK. The ABCs of accessory breast
tissue: basic information every radiologist should know. AJR Am J Roentgenol.
2014;202(5):1157-1162.
doi pubmed - Laor T, Collins MH, Emery KH, Donnelly LF, Bove KE, Ballard
ET. MRI appearance of accessory breast tissue: a diagnostic consideration for an axillary mass
in a peripubertal or pubertal girl. AJR Am J Roentgenol.
2004;183(6):1779-1781.
doi pubmed - Gou ZC, Liu XY, Xiao Y, Zhao S, Jiang YZ, Shao ZM. Decreased
survival in patients with carcinoma of axillary tail versus upper outer quadrant breast cancers:
a SEER population-based study. Cancer Manag Res. 2018;10:1133-1141.
doi pubmed - Salemis NS. Primary ectopic breast carcinoma in the axilla:
A rare presentation and review of the literature. Breast Dis. 2021;40(2):109-114.
doi pubmed - Zhang S, Yu YH, Qu W, Zhang Y, Li J. Diagnosis and treatment
of accessory breast cancer in 11 patients. Oncol Lett. 2015;10(3):1783-1788.
doi pubmed - Tjalma WA, Senten LL. The management of ectopic breast
cancer—case report. Eur J Gynaecol Oncol. 2006;27(4):414-416.
pubmed - Markopoulos C, Kouskos E, Kontzoglou K, Gogas G, Kyriakou V,
Gogas J. Breast cancer in ectopic breast tissue. Eur J Gynaecol Oncol.
2001;22(2):157-159.
pubmed - Cogswell HD, Czerny EW. Carcinoma of aberrant breast of the
axilla. Am Surg. 1961;27:388-390.
pubmed - Friedman-Eldar O, Melnikau S, Tjendra Y, Avisar E. Axillary
reverse lymphatic mapping in the treatment of axillary accessory breast cancer: a case report
and review of management. Eur J Breast Health. 2022;18(1):1-5.
doi pubmed - Madej B, Balak B, Winkler I, Burdan F. Cancer of the
accessory breast—a case report. Adv Med Sci. 2009;54(2):308-310.
doi pubmed - Hallam S, Aggarwal A, Predolac D, Cunnick G, Ashford R.
Primary ectopic breast carcinoma in a supernumerary breast arising in the anterior chest wall: a
case report and review of the literature. J Surg Case Rep. 2013;2013(12):rjt107.
doi pubmed - Hughes KS, Schnaper LA, Bellon JR, Cirrincione CT, Berry DA,
McCormick B, Muss HB, et al. Lumpectomy plus tamoxifen with or without irradiation in women age
70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin
Oncol. 2013;31(19):2382-2387.
doi pubmed - Kunkler IH, Williams LJ, Jack WJ, Cameron DA, Dixon JM,
investigators PI. Breast-conserving surgery with or without irradiation in women aged 65 years
or older with early breast cancer (PRIME II): a randomised controlled trial. Lancet Oncol.
2015;16(3):266-273.
doi pubmed - Polce S, Noldner C, Ailawadi S, Ryu S, Stessin A. Radiation therapy in cancer of accessory breast tissue: questions for treatment guidelines, case series, and literature review. Appl Radiat Oncol. 2023;(1):31-37.
- Nihon-Yanagi Y, Ueda T, Kameda N, Okazumi S. A case of
ectopic breast cancer with a literature review. Surg Oncol. 2011;20(1):35-42.
doi pubmed - Marshall MB, Moynihan JJ, Frost A, Evans SR. Ectopic breast
cancer: case report and literature review. Surg Oncol. 1994;3(5):295-304.
doi pubmed
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Medical Cases is published by Elmer Press Inc.