Figures
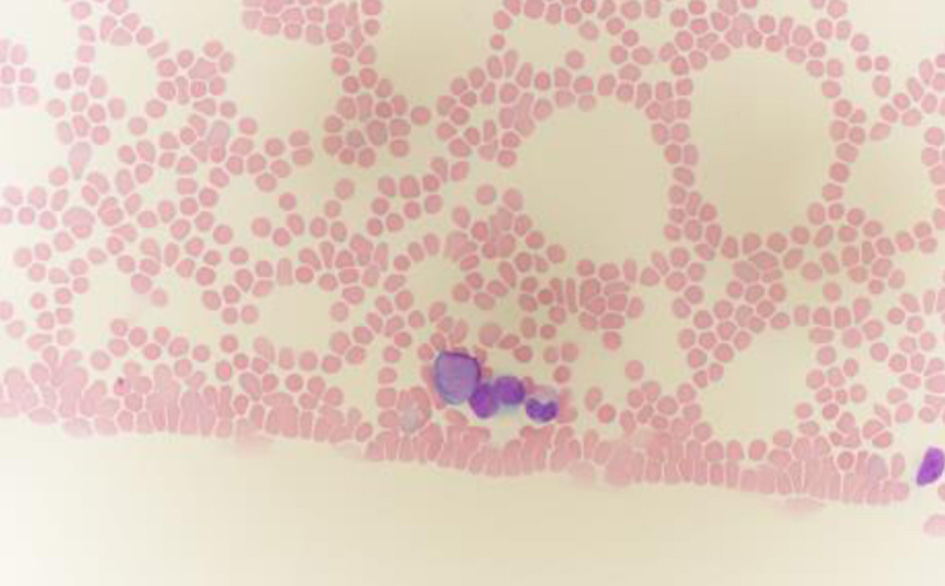
↓ Figure 1. Peripheral blood smear stained with Wright–Giemsa, showing blasts with scant blue cytoplasm (oil immersion, × 1,000). Note the characteristic features of blasts: moderate cell size, high nuclear-to-cytoplasmic ratio, fine chromatin pattern, and inconspicuous nucleoli.
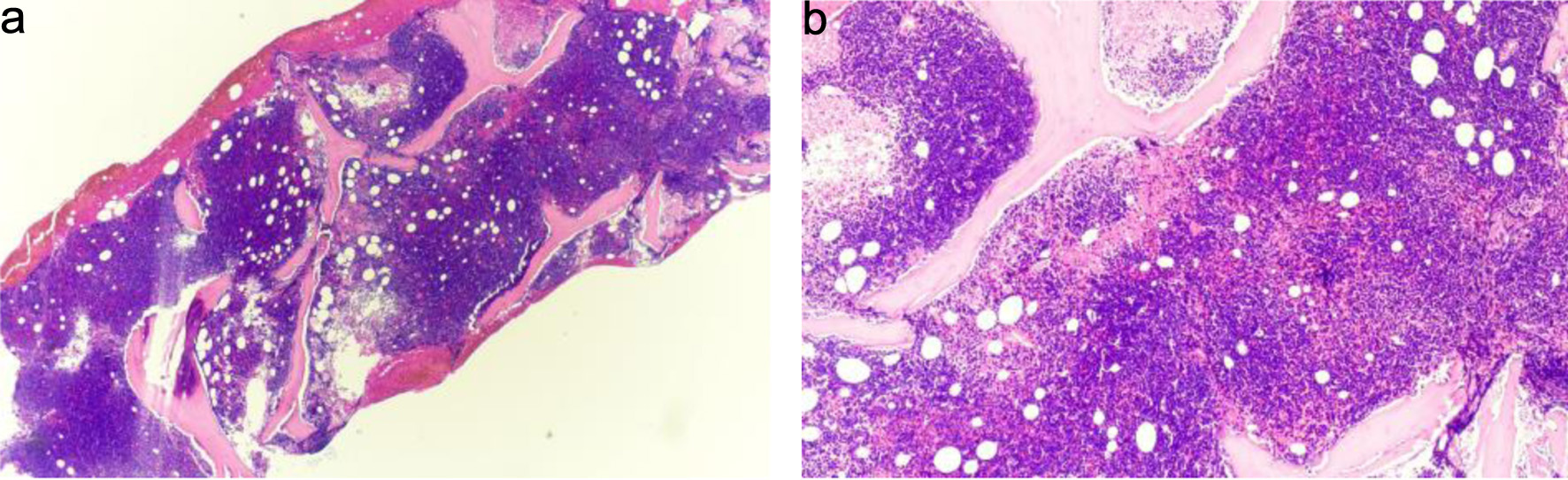
↓ Figure 2. (a) Bone marrow biopsy, hypercellular for age with diffuse replacement by blasts (hematoxylin and eosin stain, × 20). Observe the dense, monotonous cellularity with near-complete effacement of normal hematopoietic elements and marrow architecture. (b) Higher-power view showing sheets of small blue cells consistent with blasts (hematoxylin and eosin stain, × 40). The blasts demonstrate high nuclear-to-cytoplasmic ratio, scant basophilic cytoplasm, finely dispersed chromatin, and inconspicuous nucleoli—features requiring immunophenotyping for definitive lineage assignment. Immunophenotyping by flow cytometry confirmed B-lymphoid lineage (Figs. 4 and 5).
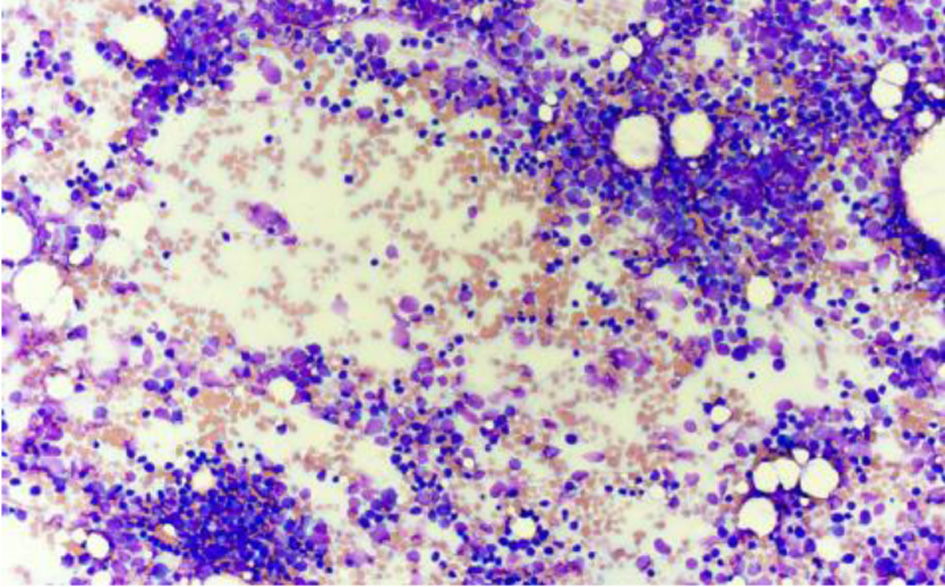
↓ Figure 3. Bone marrow aspirate smear showing increased blast population at low power (Wright–Giemsa stain, × 20). Blasts predominate throughout the aspirate smear with minimal normal hematopoietic precursors visible.
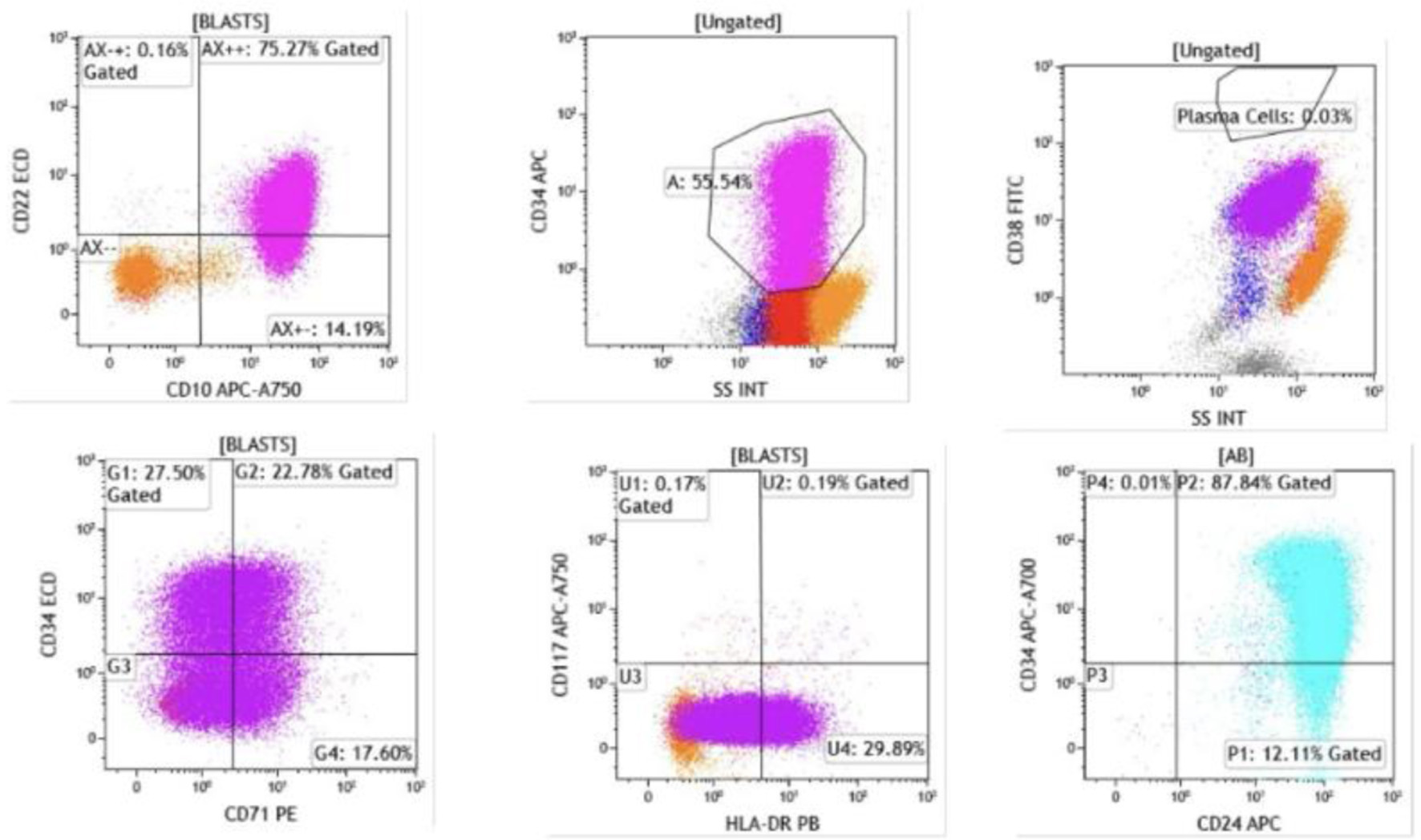
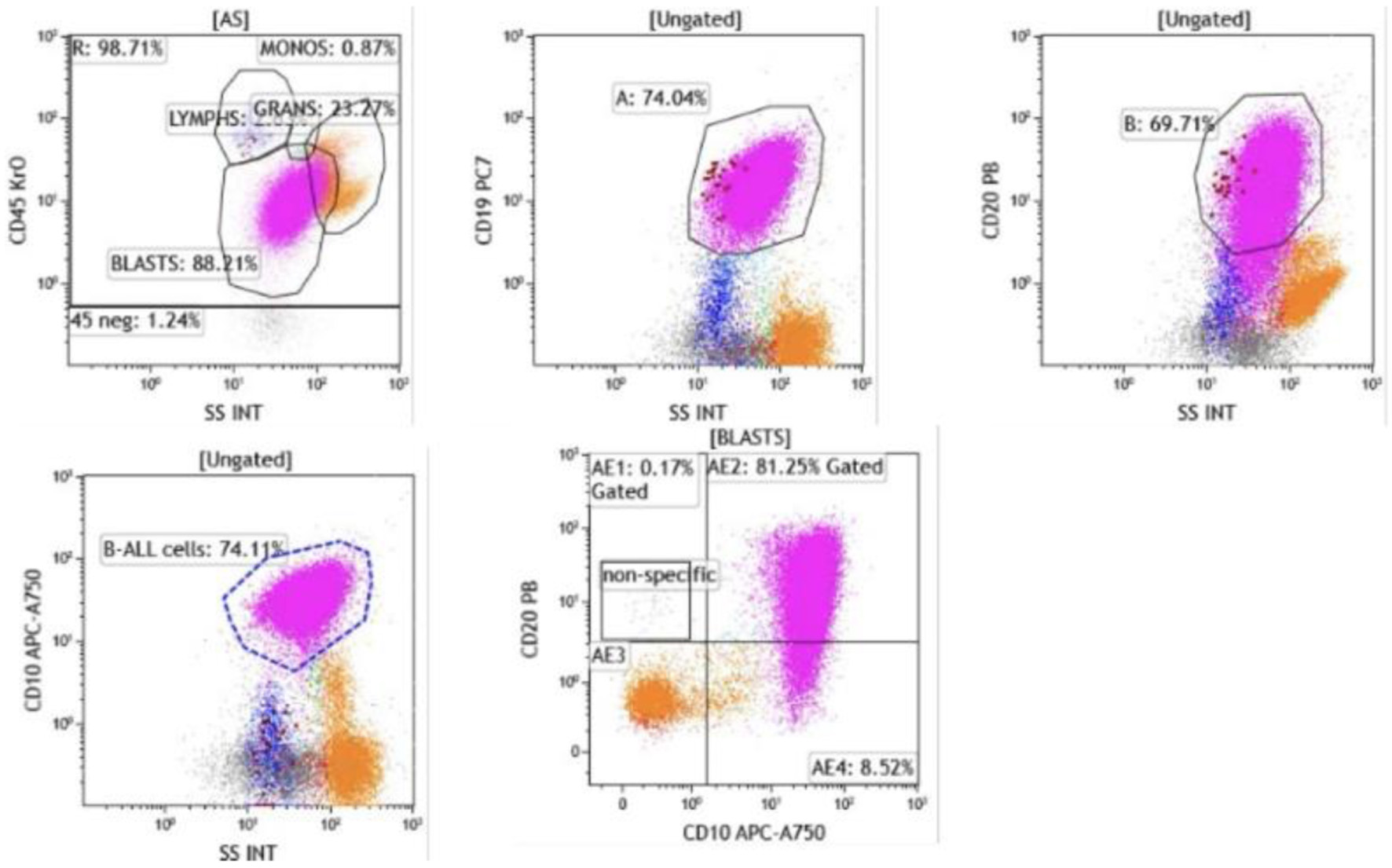
↓ Figure 4. Flow cytometry of bone marrow aspirate. Blasts were identified in the CD45-dim, low side scatter gate, comprising approximately 74% of total events. The blast population expressed CD19, CD10, and CD20 (heterogeneous, moderate to bright), confirming B-lymphoid lineage (CD19, CD10) with early precursor features.
↓ Figure 5. Flow cytometry demonstrated that blasts were positive for CD22 (dim), CD24, CD34 (heterogeneous), CD38, CD45 (dim), CD71, and HLA-DR (heterogeneous). Intracytoplasmic expression of CD79a and terminal deoxynucleotidyl transferase (TdT) was also detected. The presence of TdT and CD34 expression confirms the immature blast phenotype, while CD79a positivity further supports B-cell lineage commitment.