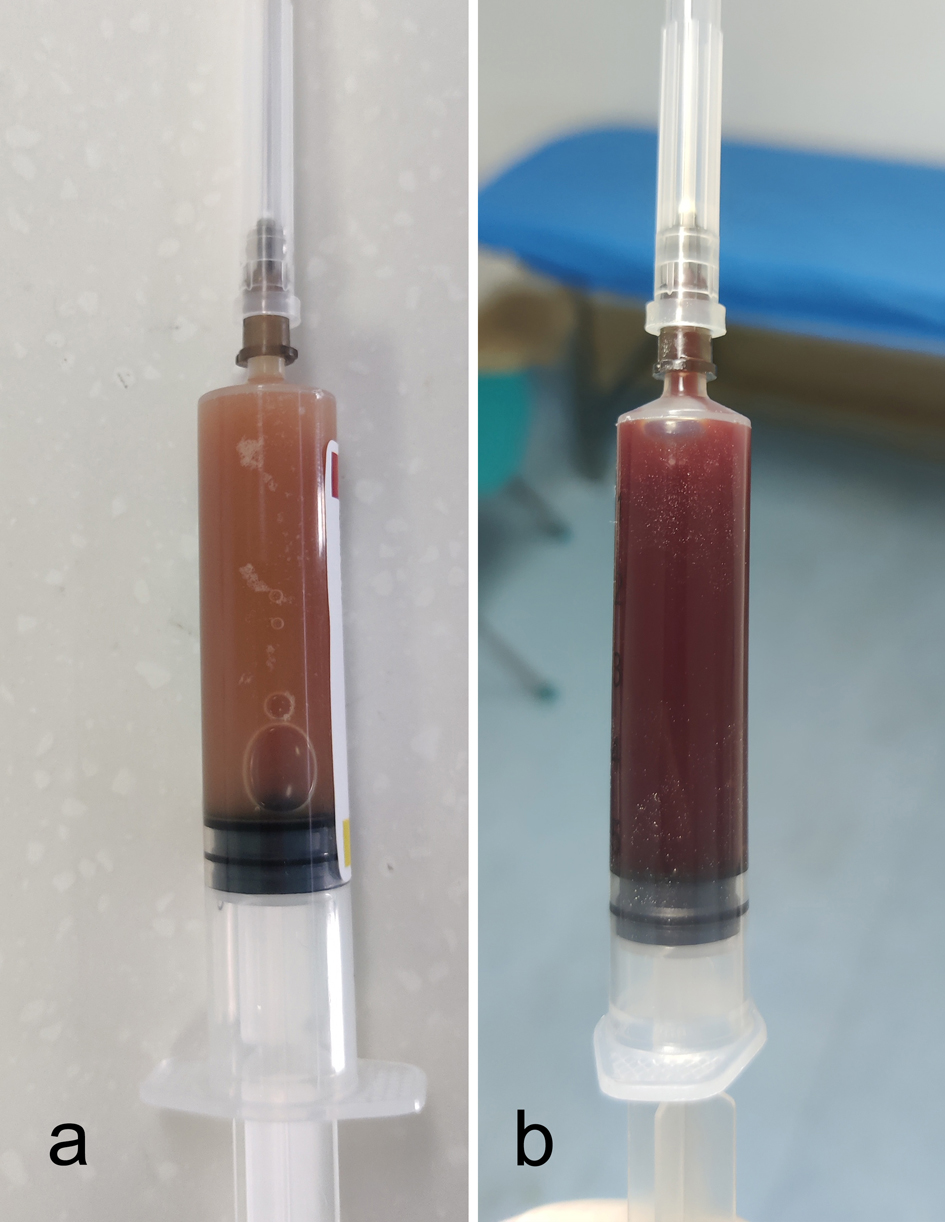
↓ Figure 1. The joint fluid of two infected
patients through knee arthrocentesis. (a) Case 1. (b): Case 2.
| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://jmc.elmerpub.com |
Case Report
Volume 16, Number 7, July 2025, pages 239-247
Investigation and Management of a Small Outbreak of Septic Knee Arthritis Following Knee Arthroscopy by Stenotrophomonas maltophilia
Figures

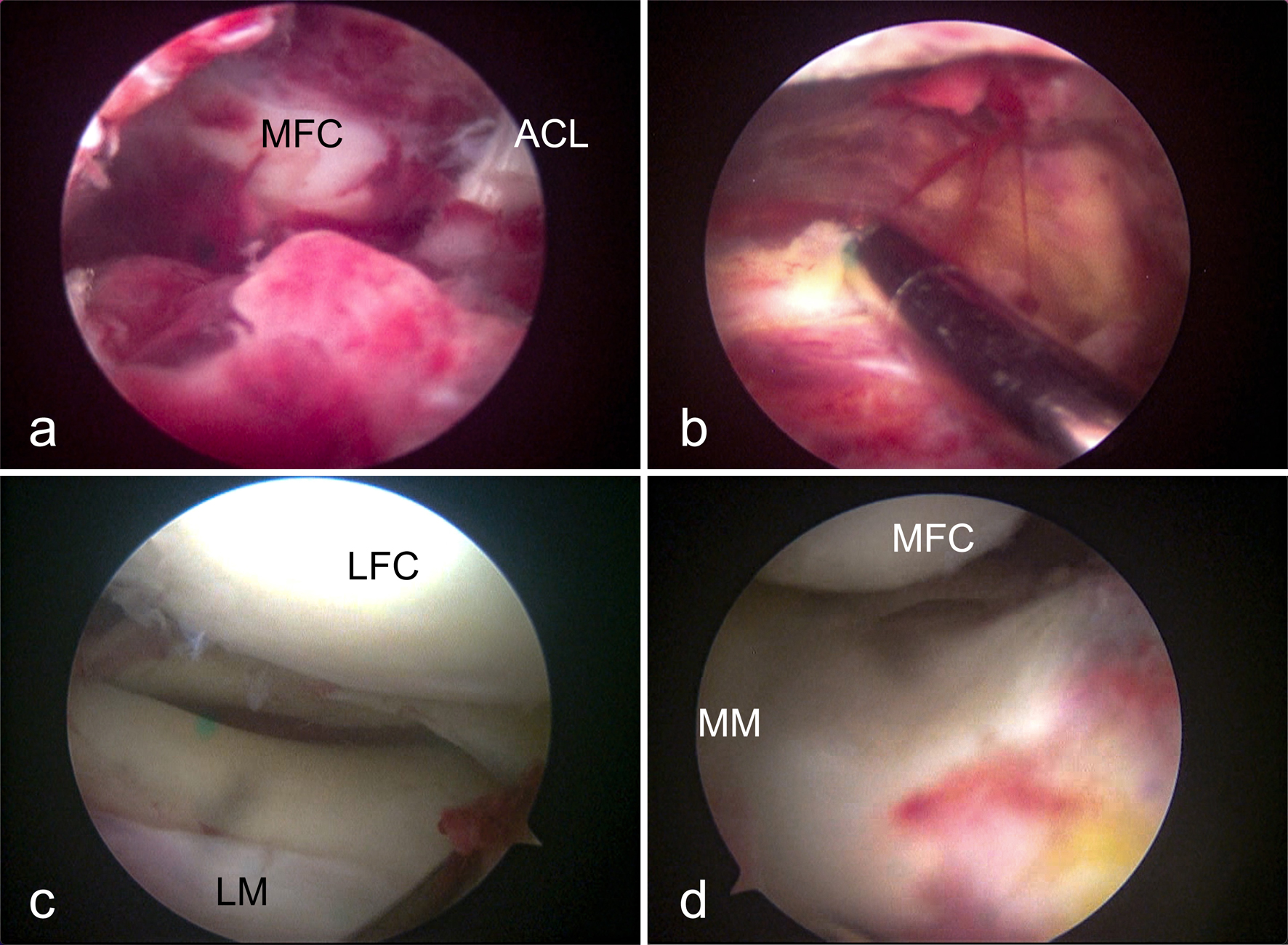
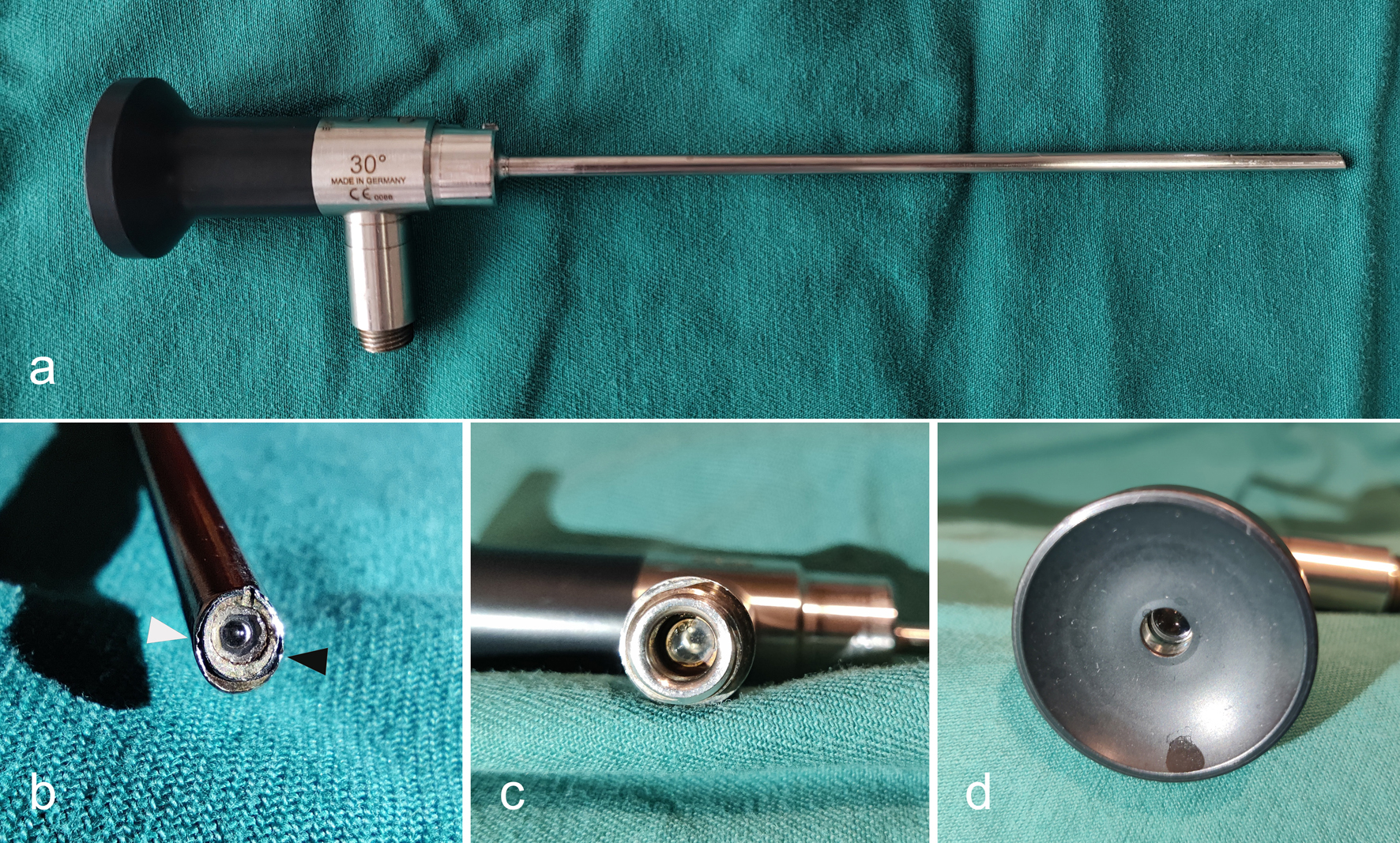
Tables
| Instrument type | Items | Sterilization | Samples per item | Sample positions | Positive samples | Positive positions |
|---|---|---|---|---|---|---|
| One to four samples per item were collected depending on instrument size, shape, and complexity. Initial test results were presented in the table. Two subsequent rounds of testing were performed, showing negative results. ACL: anterior cruciate ligament; HPGP: hydrogen peroxide gas plasma. | ||||||
| Arthroscope | 4 | HPGP | 3 | Surface, object glasses, light cable port | 1 | Object glasses |
| Shaver handpiece | 3 | HPGP | 4 | Body surface, body cavity, cable surface, plug | 0 | N/A |
| Light cable | 3 | HPGP | 3 | Cable surface, arthroscope port, light source port | 0 | N/A |
| Arthroscope cannula | 4 | Steam autoclave | 2 | Surface, cavity | 0 | N/A |
| Cannula obturator | 4 | Steam autoclave | 1 | Surface | 0 | N/A |
| Probe | 3 | Steam autoclave | 1 | Surface | 0 | N/A |
| Basket forceps | 12 | Steam autoclave | 1 | Surface | 0 | N/A |
| Grasping forceps | 6 | Steam autoclave | 1 | Surface | 0 | N/A |
| Microfracture pick | 6 | Steam autoclave | 1 | Surface | 0 | N/A |
| Arthroscopic curette | 3 | Steam autoclave | 1 | Surface | 0 | N/A |
| Suture hook | 6 | Steam autoclave | 1 | Surface | 0 | N/A |
| Suture cutter | 3 | Steam autoclave | 1 | Surface | 0 | N/A |
| Knot pusher | 3 | Steam autoclave | 1 | Surface | 0 | N/A |
| ACL femoral aimer | 2 | Steam autoclave | 1 | Surface | 0 | N/A |
| ACL tibial guide | 2 | Steam autoclave | 1 | Surface | 0 | N/A |
| ACL reamer | 12 | Steam autoclave | 1 | Surface | 0 | N/A |
| Tendon harvester | 2 | Steam autoclave | 1 | Surface | 0 | N/A |
| Graft sizer | 10 | Steam autoclave | 1 | Surface | 0 | N/A |
| Ruler | 6 | Steam autoclave | 1 | Surface | 0 | N/A |
| General surgical instruments (e.g., scalpel handle, scissor, forceps) | 56 | Steam autoclave | 1 | Surface | 0 | N/A |
| Operation type | Patients | Infected patients (positive microbiology) | Infected patients (negative microbiology) | Infection ratio |
|---|---|---|---|---|
| ACLR: anterior cruciate ligament reconstruction. | ||||
| Partial meniscectomy | 4 | 1 | 2 | 75% |
| Meniscus repair | 3 | 2 | 0 | 66.7% |
| ACLR | 8 | 1 | 0 | 12.5% |
| Synovectomy | 2 | 0 | 1 | 50.0% |
| Elbow release | 1 | 0 | 0 | 0.0% |
| Iliotibial band release | 1 | 0 | 0 | 0.0% |
| Total | 19 | 4 | 3 | 36.8% |
| Operation date | Case 1 | Case 2 | Case 3 | Case 4 |
|---|---|---|---|---|
| Day -17* | Day 4 | Day 2 | Day 5 | |
| The date of case 1’s postoperative outpatient revisit when septic knee arthritis was suspected, was defined as day 0. *Day -17 indicates 17 days before day 0. SMZ-TMP: sulfamethoxazole-trimethoprim. | ||||
| Protocol | Cefepime-sulbactam (3.0 g/8 h) | Ceftriaxone (2 g/24 h) SMZ-TMP (0.96 g/12 h) |
Ceftriaxone (2.0 g/24 h) | Levofloxacin (0.75 g/d) SMZ-TMP (1.44 g/8 h) |
| Start date | Day 1 | Day 9 | Day 6 | Day 12 |
| End date | Day 6 | Day 10 | Day 8 | Day 22 |
| Protocol | Levofloxacin (0.5 g/day) SMZ-TMP (0.96 g/8 h) |
Levofloxacin (0.75 g/day) SMZ-TMP (1.44 g/8 h) |
Levofloxacin (0.75 g/day) SMZ-TMP (1.44 g/8 h) |
SMZ-TMP (1.44 g/8 h) Minocycline (100 mg/12 h) |
| Start date | Day 7 | Day 11 | Day 9 | Day 23 |
| End date | Day 11 | Day 17 | Day 22 | Day 50 |
| Protocol | Levofloxacin (0.75 g/day) SMZ-TMP (1.44 g/8 h) Minocycline (100 mg/12 h) |
SMZ-TMP (1.44 g/8 h) Minocycline (100 mg/12 h) |
SMZ-TMP (1.44 g/8 h) Minocycline (100 mg/12 h) |
|
| Start date | Day 12 | Day 18 | Day 23 | |
| End date | Day 67 | Day 46 | Day 50 | |