| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://jmc.elmerpub.com |
Case Report
Volume 16, Number 3, March 2025, pages 102-106
Combination of 2-Chlorodeoxyadenosine, Cytarabine, and Granulocyte Colony-Stimulating Factor and Venetoclax in a Case of Acute Myelogenous Leukemia
Ella Prebel Jackerta, Karrune Woanb, Preet Chaudharyb, George Yaghmourb, Abdullah Ladhab, Eric Tamb, c
aKeck School of Medicine, University of Southern California, Los Angeles, CA 90089,
USA
bDepartment of Bone and Marrow Transplant, USC Norris Comprehensive Cancer
Center, Los Angeles, CA 90089, USA
cCorresponding Author: Eric Tam, Department
of Bone and Marrow Transplant, USC Norris Comprehensive Cancer Center, Los Angeles, CA 90089,
USA
Manuscript submitted November 21, 2024, accepted January 23, 2025, published online February 18,
2025
Short title: CLAG + VEN in Refractory AML
doi: https://doi.org/10.14740/jmc5087
| Abstract | ▴Top |
The high rates of relapse following induction therapy for acute myelogenous leukemia (AML) warrant the investigation of novel chemotherapy regimens to better treat the disease safely. We report a case of refractory AML treated with CLAG (a combination of 2-chlorodeoxyadenosine, cytarabine, and granulocyte colony-stimulating factor (GCSF)), as a replacement for FLAG-IDA (fludarabine, cytarabine, G-CSF and idarubicin), due to a shortage of fludarabine, plus B-cell lymphoma-2 (BCL-2) inhibitor venetoclax (CLAG + VEN). A 38-year-old woman with a past medical history of systemic lupus erythematosus (SLE), managed on hydroxychloroquine, presented to her primary care provider with worsening fatigue and was found to have significant leukocytosis. The patient was diagnosed with AML on bone marrow biopsy (BMBX). The patient delayed care after the initial diagnosis but eventually started on a continuous infusion of cytarabine for therapy day (D) 1 - D7 and daunorubicin 60 mg/m2 (D1 - D3) (7 + 3) induction chemotherapy. A BMBX was performed on D18 following induction therapy, revealing residual disease with 46% blasts, indicative of refractory AML. Three weeks after completing induction therapy, the patient underwent CLAG + VEN. After completing CLAG + VEN, she was found to be minimal residual disease (MRD)-negative and was determined to be an appropriate candidate for bone marrow transplant (BMT) following maintenance therapy with Onureg (azacitidine). The patient successfully underwent BMT and remains MRD-negative 1 year post-transplant. Treatment with CLAG + VEN was effective in achieving remission in this case, enabling this patient to successfully undergo BMT. This suggests a potential therapeutic benefit of combining venetoclax with traditional CLAG therapy in complex cases of AML.
Keywords: Acute myelogenous leukemia; Induction chemotherapy; Bone marrow transplant; CLAG; Venetoclax
| Introduction | ▴Top |
Due to the aggressive nature of relapsed/refractory acute myelogenous leukemia (AML), a combination chemotherapy regimen is often administered. According to the National Comprehensive Cancer Network guidelines, relapse after induction therapy with the antimetabolite cytarabine plus an anthracycline, such as idarubicin (IDA) or daunorubicin (7 + 3), warrants a secondary aggressive chemotherapy regimen, typically involving high-dose cytarabine (HiDAC)-based therapy. Although this treatment paradigm has been shown to be effective in achieving complete remission in some cases, long-term survival remains limited to only 30-40% following standard chemotherapy [1]. Thus, the discovery and implementation of new chemotherapy drugs and regimens specifically for relapsed/refractory AML are of utmost importance.
With eligibility for hematopoietic stem cell transplant (HSCT) being the end-goal, recent new drug combinations have been shown to be both safe and effective in treating relapsed/refractory AML. One recommended multi-agent regimen consisting of fludarabine, cytarabine, granulocyte colony-stimulating factor (G-CSF), and IDA (FLAG-IDA), in combination with B-cell lymphoma-2 (BCL-2) inhibitor venetoclax (VEN), has demonstrated reduced relapse rates and increased remission, with a high rate of successful transition to HSCT [2-5]. Due to a national shortage of fludarabine in 2022, we substituted the FLAG-IDA combination with the cladribine, cytarabine, G-CSF (CLAG) regimen, in combination with VEN, for treatment in this case. The CLAG induction regimen has been successful in achieving complete remission on its own [6]. Due to the aggressive presentation and poor prognostic cytogenetics of this patient, and understanding of the success of FLAG + VEN in other cases, VEN was included in the treatment protocol. To the best of our knowledge, this is one of the first reported cases using CLAG + VEN for the treatment of relapsed/refractory AML.
| Case Report | ▴Top |
Investigations
A 38-year-old woman with a past medical history of systemic lupus erythematosus (SLE), managed with hydroxychloroquine, presented to her primary care provider with worsening fatigue. Routine labs revealed significant leukocytosis.
Diagnosis
The patient was referred to hematology and underwent a bone marrow biopsy (BMBX), which revealed 57% blasts, leading to a diagnosis of AML. Cytogenetic analysis was negative for FLT3, TP53, NPM1, CEBPA, t(8:21), inv(16), t(15:17), and BCR-ABL1. The patient denied family history of malignancy. The patient delayed care after the initial diagnosis, but eventually started on induction chemotherapy with a continuous infusion of cytarabine (day (D) 1 - D7) and daunorubicin 60 mg/m2 (D1 - D3) (7 + 3). The patient’s hospitalization for induction therapy was complicated by a culture-negative neutropenic fever.
A repeat BMBX was performed on D18 following induction therapy, revealing residual disease with 46% blasts, indicative of refractory AML. The patient was transferred to a higher level of care for re-induction 3 weeks after completing induction therapy. Physical exam was unremarkable upon admission; however, the patient presented with leukopenia, anemia, and thrombocytosis (white blood cell (WBC) 1.2 × 103 cells/µL (normal range 4.00 - 10.90 × 103 cells/µL), hemoglobin (Hgb) 9.1 g/dL (normal range 12.3 - 15.3 g/dL), hematocrit 27.1% (normal range 36-48%) , platelet (PLT) count 510 × 103 cells/µL (normal range 150 - 400 × 103 cells/µL)). FLAG + VEN was initially planned as salvage therapy, however, due to the fludarabine shortage, the regimen was changed to CLAG. VEN was added in the hopes of achieving a quicker and deeper remission.
Treatment
The induction therapy regimen was as follows: VEN 400 mg/day orally (PO) (D0 - D14), filgrastim 300 µg/day (D1 - D5), cladribine 5 mg/m2/day intravenous (IV) infusion (D1 - D5), cytarabine 2 mg/m2/day IV infusion over 4 h, initiated 2 h following cladribine infusion (D1 - D5). Zarxio (filgrastim-sndz) was continued after chemotherapy for blood count recovery.
Overall, the patient tolerated therapy well. During the infusions, the patient experienced mild intermittent nausea (grade 1), which was controlled as needed with Zofran (ondansetron). The initial presenting leukocytosis of 51.58 × 103 cells/µL decreased rapidly during chemotherapy, progressing to neutropenia. The patient developed menorrhagia, which was treated with Aygestin (norethindrone acetate) and Amicar (aminocaproic acid), but ultimately required packed red blood cell (PRBC) transfusions due to light-headedness and abdominal pain. Culture-negative neutropenic fever resolved with empirical antibiotics. The patient underwent a repeat BMBX on D27 of CLAG + VEN, which demonstrated complete remission with blasts less than 5% and no irregular myeloid progenitor cells, indicating negative minimal residual disease (MRD) by multicolor flow cytometry. G-CSF was restarted for primary prophylaxis, which had delayed success in count recovery; however, the patient continued to experience prolonged thrombocytopenia.
The patient was discharged from the hospital on D39 after resolution of fever and menorrhagia, but continued with grade 4 thrombocytopenia (based on Common Terminology Criteria for Adverse Events (CTCAE) 5.0). She was started on Promacta (eltrombopag) in an effort to achieve PLT count recovery. The patient continued to return to the clinic weekly for follow-up and ongoing evaluation for bone marrow transplant (BMT).
Following completion of CLAG + VEN induction therapy, there were < 5% blast cells on BMBX, MRD negative, and normocellular marrow, with no abnormal myeloid progenitor population identified. The patient underwent one round of consolidation therapy with HiDAC for 6 days, starting 76 days after CLAG + VEN induction therapy. The patient was then transitioned to maintenance therapy with Onureg (azacitidine) and received three doses of romiplostim 4 µg/kg for ongoing management of thrombocytopenia during this period.
Follow-up and outcomes
The patient remained MRD-negative and became a candidate for BMT after completing approximately four 14-day cycles of Onureg for maintenance therapy. The transplant donor was a 23-year-old female, blood type O+, cytomegalovirus (CMV) negative, and haploidentical. The patient then underwent myeloablative conditioning with fractionated total body irradiation (FTBI) and fludarabine prior to the transplant. Post-transplant graft-vs-host disease (GVHD) prophylaxis included post-transplant cyclophosphamide and tacrolimus-mycophenolate mofetil, along with letermovir for CMV prophylaxis due to T lymphocyte depletion and the increased risk of invasive CMV disease following BMT. BMT was complicated by GVHD affecting the skin and upper gastrointestinal (GI) tract, resulting in rashes, decreased oral intake, and weight loss in the postoperative period. The patient was started on prednisone up to 1 mg/kg, followed by a slow taper, but continued to experience persistent GVHD symptoms, including upper GI involvement (mouth) and likely the musculoskeletal system, with ongoing muscular aches and pains. As a result, management was transitioned to Janus kinase-2 (JAK-2) inhibitor Jakafi (ruxolitinib) 5 mg and Rho-associated kinase 2 (ROCK-2) inhibitor Rezurock (belumosudil) 200 mg. The patient was CMV-positive prior to transplant and so remained on acyclovir 800 mg two times a day (BID) for prophylaxis of reactivated CMV disease. Additionally, Cresemba (isavuconazonium sulfate) and Bactrim (trimethoprim-sulfamethoxazole) were used for antifungal and antibacterial disease prophylaxis, respectively.
Interval positron emission tomography-computed tomography (PET-CT) scans demonstrated hypermetabolic activity in the inguinal lymph nodes; however, these lymph nodes were < 1 cm in size, and thus, were not biopsied. These nodes remained stable on subsequent PET studies. Interval BMBX following BMT continued to demonstrate MRD-negative status. One year following BMT, the patient underwent PET-CT, BMBX and chimerism studies. The PET-CT showed decreased hypermetabolic activity in the previously noted lymph nodes, with no new areas of hypermetabolic activity. New, nonspecific patchy bilateral upper-lobe predominant ground-glass opacities of the lungs were identified, likely reflecting infectious or inflammatory processes, but were not concerning for metastatic disease. The BMBX was again MRD-negative, with no blasts or abnormal myeloid progenitor cells identified. Chimerism study confirmed full engraftment. These findings suggested that this patient was in complete remission from leukemia at 1 year post-BMT.
| Discussion | ▴Top |
Here, we present a case of refractory AML treated with a combination therapy of CLAG + VEN. The patient was initially treated with the classic 7 + 3 induction regimen, but unfortunately, the disease persisted. After transfer of care to a tertiary center, FLAG + VEN was initially planned; however, it was switched to CLAG + VEN due to a national shortage of fludarabine. CLAG therapy has been shown to be an effective induction treatment for relapsed/refractory AML, and VEN was added to this regimen in the hopes of achieving a quicker and deeper remission. This treatment was successful in achieving remission, with a BMBX on D27 of CLAG + VEN showing MRD-negative status.
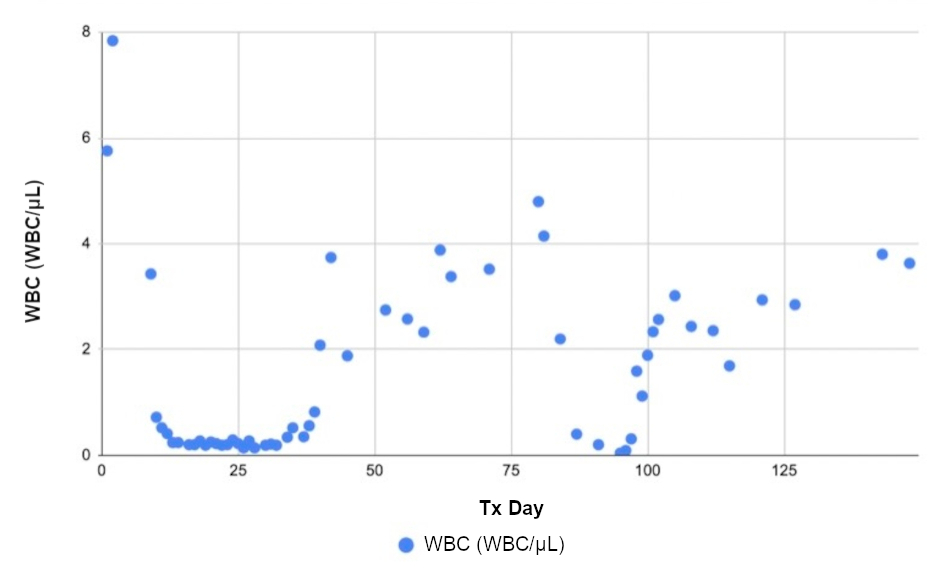
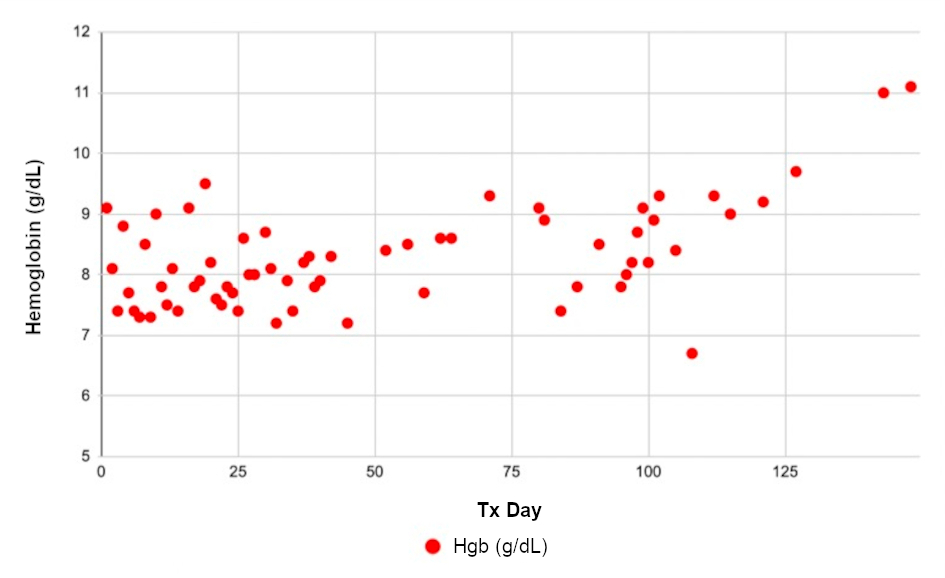
The patient’s blood counts have been continuously monitored. Trends in WBC count, Hgb, and PLT levels are shown in Figures 1, 2 and 3. Over the course of 3 months following treatment, the WBC count (Fig. 1) and Hgb (Fig. 2) have trended upwards, suggesting sustained remission and delayed hematopoietic recovery. A decrease in all blood counts is noted on D76 - 81, corresponding to HiDAC consolidation therapy, after which blood counts resumed an upward trend toward recovery.
 Click for large image |
Figure 1. White blood cell (WBC) counts as a function of treatment (Tx) day. |
 Click for large image |
Figure 2. Hemoglobin (Hgb) as a function of treatment (Tx) day. |
 Click for large image |
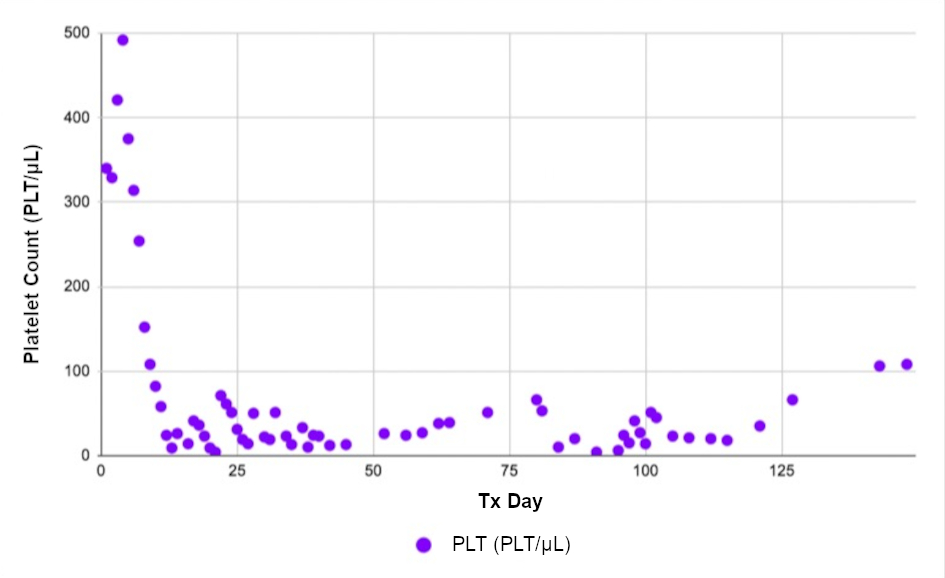
Figure 3. Platelet (PLT) counts as a function of treatment (Tx) day. |
The PLT count trended downwards overall (Fig. 3), in contrast to WBC and Hgb. There was a slight increase in PLT count after completion of CLAG + VEN, but it was not significant enough to be considered recovery. PLT count fluctuated between 4,000 and 50,000 for the 3 months following therapy, with a decrease observed on D76 - 81, consistent with the effects of HiDAC consolidation therapy. Prior to CLAG + VEN therapy, the patient’s PLT count was 340,000. More recent lab results showed an increase in PLT count to 101,000, measured on D148, following initiation of CLAG + VEN therapy. The patient’s PLT count has since remained above 100,000.
In addition to the hematologic adverse effect of prolonged thrombocytopenia, which continues to be monitored, other adverse events were noted. The patient suffered from culture-negative neutropenic fever, grade 1 nausea, abdominal pain, and menorrhagia, which required PRBC transfusions. Many of these adverse effects are comparable to those reported in previous studies of CLAG and FLAG + VEN treatment. An early study by Robak et al evaluating the efficacy and safety of the CLAG regimen in 20 AML patients reported adverse effects such as pancytopenia, infection complications, pulmonary edema, heart failure and diarrhea [6]. However, the study also noted that the median time to achieve PLT count recovery was 27.2 days. Similarly, a study conducted by DiNardo et al evaluating the efficacy and safety of the FLAG + VEN combination treatment in 68 AML patients found adverse effects including neutropenia, bacteremia, pneumonia, sepsis, abdominal pain and GI disorders [1]. The median time to achieve PLT count recovery was 31 - 37 days, depending on the treatment group. Many of the adverse effects observed in this case align with findings from CLAG and FLAG + VEN treatment regimens. Notable differences include prolonged thrombocytopenia, which failed to recover for 148 days, and menorrhagia requiring PRBC transfusions. Moreover, cardiac and pulmonary complications, such as heart failure, pulmonary edema and pneumonia were not noted in our case. The study of Robak et al also reported that the majority of patients on CLAG therapy alone relapsed after induction, despite intensive consolidation and maintenance therapy [6]. The MRD-negative outcome and eventual successful BMT in our case supports an additional therapeutic benefit of adding VEN to CLAG induction therapy for refractory AML, compared to CLAG alone.
A published abstract by Aifuwa et al, presented at the American Society of Hematology, described two cases of CLAG + VEN therapy for relapsed/refractory AML, treated late in their disease course, who both ultimately died from relapse [7]. One case involved a patient with an identified FLT3-integral tandem duplication (FLT3-ITD) mutation, who underwent cytarabine and mitoxantrone followed by HiDAC. However, the patient relapsed 5 months after treatment and was subsequently started on CLAG + VEN. This patient had multiple gene mutations, including FLT3-ITD, DNMT3A, NPM1, and WT1, whereas the patient we present had no identified mutations at any point in the disease course. The authors did not report the date of death after relapse for the first case. The second case involved a 71-year-old female with 45 XX add(5)(q11), der(6:7)(p10;q10) karyotype and NRAS mutation. This patient received CLAG-IDA induction chemotherapy followed by HSCT, relapsed after transplant, and was subsequently treated with CLAG + VEN as salvage therapy. This patient’s disease was complicated by the recurrence of extramedullary lymph node disease and continued to progress, ultimately leading to death on D381 post-transplant. These cases, along with the one we present here, demonstrate three examples of CLAG + VEN used as salvage therapy for relapsed/refractory AML, with each patient having a distinct cytologic disease profile. Neither of the previously published cases used CLAG + VEN as a bridge to HSCT, as we did in our case; therefore, they are not easily comparable to the use of CLAG + VEN in our case. Additionally, these studies did not report blood cell count outcomes of the patients, which makes it difficult to determine if our finding of prolonged thrombocytopenia is observed in other cases. Thus, further studies are warranted with consistent therapy regimens using CLAG + VEN to better discern consistent long-term outcomes for this patient population.
In conclusion, CLAG + VEN combination therapy for relapsed/refractory AML is a safe and efficacious substitute for FLAG +VEN chemotherapy, and may have increased therapeutic benefits for achieving remission compared to CLAG therapy administered without VEN. These findings encourage future studies, particularly clinical trials, to investigate modified and novel chemotherapy regimens for AML.
Learning points
CLAG + VEN combination therapy shows promise as a safe and efficacious treatment for relapsed/refractory AML, highlighting its potential in clinical practice.
Prolonged thrombocytopenia following CLAG + VEN therapy warrants further exploration, as it could impact patient management and outcomes, indicating an important area for clinical focus.
The long-term survival outcomes following CLAG + VEN therapy remain an area for future investigation, suggesting that monitoring long-term effects will be crucial for optimizing care and improving patient prognosis.
Acknowledgments
We acknowledge the University of Southern California (USC) Norris Comprehensive Cancer Center and the USC Norris Bone and Marrow Transplant Center for continued commitment to advancing patient care through research.
Financial Disclosure
No funding was received for this study.
Conflict of Interest
None to declare.
Informed Consent
Consent was obtained to utilize deidentified patient information from the patient involved in this case prior to the submission of this case report.
Author Contributions
Ella Prebel Jackert (first author) contributed to data collection, data analysis, manuscript writing and editing. Karrune Woan, Preet Chaudhary and George Yaghmour contributed to the editing of the manuscript. Abdullah Ladha served as an editor and participated in patient care. Eric Tam was the senior author and principal investigator, participated in patient care, and contributed as an editor.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
AML: acute myelogenous leukemia; CLAG: 2-chlorodeoxyadenosine, cytarabine, and granulocyte colony-stimulating factor; FLAG: fludarabine, cytarabine, granulocyte colony-stimulating factor; VEN: venetoclax; SLE: systemic lupus erythematosus; BMBX: bone marrow biopsy; MRD: minimal residual disease; BMT: bone marrow transplant; HSCT: hematopoietic stem cell transplant; IV: intravenous; PRBC: packed red blood cell; HiDAC: high-dose cytarabine; CMV: cytomegalovirus; GVHD: graft-vs-host disease; GI: gastrointestinal; PLT: platelet; WBC: white blood cell; PET-CT: positron emission tomography-computed tomography; ITD: integral tandem duplication; D: day; NPM1: nucleophosmin; FLT3: FMS-like tyrosine kinase 3; IDA: idarubicin
| References | ▴Top |
- DiNardo CD, Lachowiez CA, Takahashi K, Loghavi S, Xiao L,
Kadia T, Daver N, et al. Venetoclax combined with FLAG-IDA induction and consolidation in newly
diagnosed and relapsed or refractory acute myeloid leukemia. J Clin Oncol.
2021;39(25):2768-2778.
doi pubmed - Pollyea DA, Amaya M, Strati P, Konopleva MY. Venetoclax for
AML: changing the treatment paradigm. Blood Adv. 2019;3(24):4326-4335.
doi pubmed - Bose P, Vachhani P, Cortes JE. Treatment of
relapsed/refractory acute myeloid leukemia. Curr Treat Options Oncol. 2017;18(3):17.
doi pubmed - Burnett AK, Russell NH, Hills RK, Hunter AE, Kjeldsen L, Yin
J, Gibson BE, et al. Optimization of chemotherapy for younger patients with acute myeloid
leukemia: results of the medical research council AML15 trial. J Clin Oncol.
2013;31(27):3360-3368.
doi pubmed - Rai KR, Holland JF, Glidewell OJ, Weinberg V, Brunner K,
Obrecht JP, Preisler HD, et al. Treatment of acute myelocytic leukemia: a study by cancer and
leukemia group B. Blood. 1981;58(6):1203-1212.
pubmed - Robak T, Wrzesien-Kus A, Lech-Maranda E, Kowal M, Dmoszynska
A. Combination regimen of cladribine (2-chlorodeoxyadenosine), cytarabine and G-CSF (CLAG) as
induction therapy for patients with relapsed or refractory acute myeloid leukemia. Leuk
Lymphoma. 2000;39(1-2):121-129.
doi pubmed - Aifuwa E, Ahmed T, Liu D, Seiter K, Steinberg A. First reported case series of combined treatment with CLAG and Venetoclax for acute myeloid leukemia (AML). Blood. 2022;140(Supplement 1):11732-11733.
This
article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0
International License, which permits unrestricted non-commercial use, distribution, and
reproduction in any medium, provided the original work is properly cited.
Journal
of Medical Cases is published by Elmer Press Inc.